Human Pulmonary Mesenchymal Stem Cells (HPMSC)
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Human Pulmonary Mesenchymal Stem Cells (HPMSC) are stem-like cells extracted from lung tissue. They are unique in that they possess the plasticity of a mesenchymal stem cell while having a lung specific identity. In the lung, HPMSCs display typical mesenchymal stem cell properties, with multipotency to fibroblasts, chondrocytes, and adipocytes, along with roles in lung tissue regeneration and modulation of inflammatory cascades.
HPMSCs in culture are spindle-shaped fibroblast-like cells that secrete many cytokines and growth factors. As such, the proliferation, secretion, and multipotency of HPMSCs make it a relevant system with which to study the regeneration and modulation of lung tissue damage, inflammatory lung conditions, and pulmonary fibrosis. HPMSCs are often used as a lung specific system to study regeneration mechanisms of injury and regeneration, stem cell differentiation and differentiation cues, as well as translational cell-based therapies for lung injuries and diseases. The plasticity, secretory nature, and reactivity to environmental cues of HPMSCs make it a good system for preclinical models to test regenerative medicine strategies, antifibrotic agents, and immunomodulatory agents.
Effect of Fe3O4@Au Nanoparticles on the Cell Viability of HPMSCs
Targeted delivery of nanoparticles to the unreachable tumors is one of the main problems in nanomedicine. Mesenchymal stem cells (MSCs) are capable of actively homing into the tumors and release the loaded antitumor agents/nanomaterials. Combining radiosensitizing agents with MSCs is a promising cancer strategy. Arcambal et al. assessed whether Fe3O4@Au nanoparticles can modulate human pulmonary MSCs (HPMSCs) to serve as carriers for radiosensitizers.
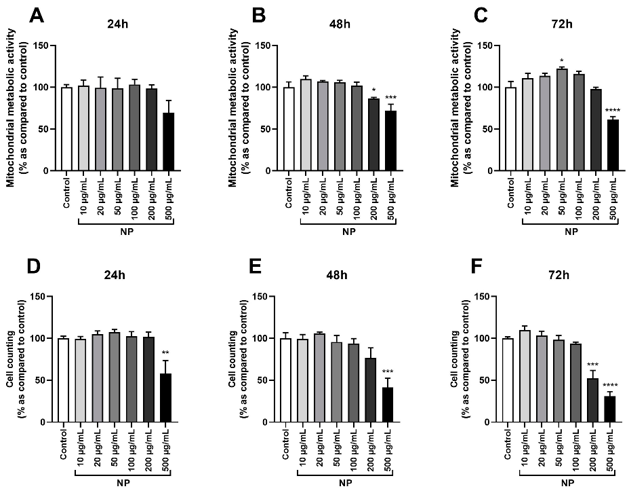
To select an appropriate Fe3O4@Au NPs concentration for further studies, mitochondrial metabolic activity and cell death were evaluated. Fe3O4@Au NPs did not have a significant effect on mitochondrial activity at 24 h, but markedly decreased it at 500 µg/mL after 48 and 72 h (Fig. 1A-C). At the same time, after 48 h of incubation, cells treated with 200 µg/mL NPs were significantly affected (Fig. 1B), and an increase in the mitochondrial activity was observed at 50 µg/mL after 72 h (Fig. 1C). Cell counting confirmed these results. Fe3O4@Au NPs significantly reduced cell viability at 500 µg/mL after 24, 48, and 72 h (Fig. 1D-F) and were toxic at 200 µg/mL after 72 h (Fig. 1F). Fe3O4@Au NPs at concentrations of 10-100 µg/mL were not significant for HPMSCs.
Ask a Question
Write your own review
- You May Also Need
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells