QualiCell® Human Amniotic Fluid Stem Cells
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Human Amniotic Fluid Stem Cells (hAFSCs) are a relatively recently discovered population of highly multi-potent stem cells thought to represent an intermediate stage between embryonic and adult stem cells. Generally collected during the second trimester by amniocentesis or with the waste amniotic fluid that accompanies birth, they express the mesenchymal marker CD117 (c-kit).
They lack the teratoma formation potential associated with embryonic stem cells and do not require destruction of a blastocyst to harvest, alleviating some of the major ethical issues. They have also been shown to have high proliferative ability and multi-differentiate into lineages from all three germ layers, including osteogenic, myogenic, adipogenic and neurogenic populations.
Recently there has been an increased focus on their paracrine signaling potential. hAFSCs secrete large amounts of bioactive molecules and exosomes with strong anti-inflammatory, pro-angiogenic, and cytoprotective effects. For these reasons hAFSC lines are becoming one of the most robust resources for cell-free and cell-based therapeutics for neonatal pathologies such as necrotizing enterocolitis as well as many inflammatory and degenerative diseases. Their ease of expansion in vitro and genetic stability are a major bonus.
Comparison of AFSC-exos and HBM-exos for the Treatment of NEC
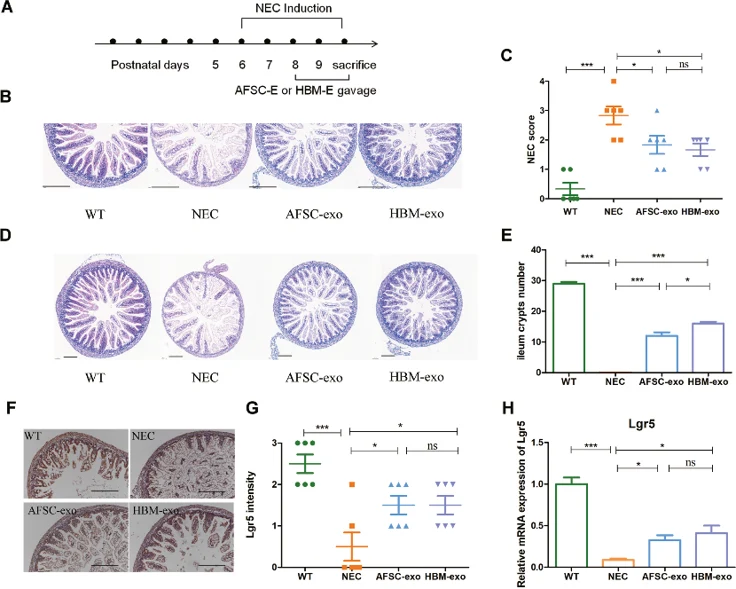
Neonatal necrotizing enterocolitis (NEC) is a devastating condition for newborns. While exosomes derived from human amniotic fluid stem cells (AFSC) and human breast milk (HBM) are known to offer protection, the specific mechanisms behind their efficacy remain unclear. Hu's team aims to compare the therapeutic effects of AFSC-derived and HBM-derived exosomes in a NEC mouse model and to identify their respective regulatory pathways in alleviating intestinal injury and inflammation.
While both AFSC-exos and HBM-exos are known to benefit NEC models, their specific molecular mechanisms remain unclear. To investigate, they established a NEC mouse model (Fig. 1A) and administered isolated exosomes. The NEC group exhibited significant intestinal injury, particularly in the ileum. Intervention with both AFSC-exos and HBM-exos effectively alleviated injury and lowered NEC scores (Fig. 1B, C). Notably, HBM-exos were more effective at restoring ileal crypt numbers than AFSC-exos (Fig. 1D, E). Furthermore, both exosome types successfully rescued the depletion of Lgr5+ intestinal stem cells and Lgr5 gene expression levels caused by NEC (Fig. 1F-H). While these treatments reduced intestinal severity, they did not significantly alter overall body weight or survival during this short-term intervention These results suggest that while both exosome sources are therapeutic, they likely operate through distinct mechanisms.
Ask a Question
Write your own review
- You May Also Need
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells