APP Transgenic Models
Aβ peptides are natural products of metabolism involving APP cleavage by β - and γ-secretase activity in the amyloidogenic pathway. Extracellular β-amyloid (Aβ) deposition is one of the most important histopathological hallmarks of Alzheimer's disease. Increased Aβ production and decreased Aβ clearance with its aggregation and deposition in intra- and extra-cellular compartments were known as the ‘Amyloid hypothesis’ for the pathogenic and neurodegenerative processes in AD. In addition, soluble aggregates, known as Aβ oligomers, have more viewed as the main toxic substance that leads to synaptic dysfunction, neurocyte apoptosis and cognitive impairment. We utilize genetic manipulation to generate APP transgenic AD model mice. The mutations involving the APP gene from which Aβ production is elevated and these transgenic mice develop neuropathological features of AD including cognitive impairment, plaque deposition in the brain and neuroinflammation. Creative Bioarray specializes in providing customized pharmacodynamic research services to help customers assess the efficacy of drug candidates and investigate the pathogenesis and progression of AD through APP transgenic models.
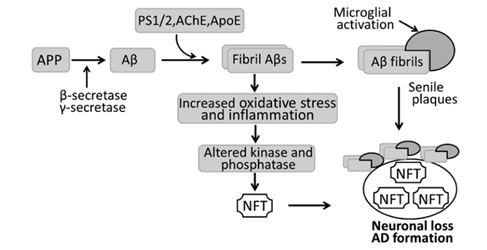 Figure. 1. Schematic diagram of the pathogenesis of Alzheimer's disease (AD). In AD, amyloid precursor protein (APP) is hydrolyzed by β-secretase and then γ-secretase into β-amyloid (Aβ) that aggregates to form fibril Aβs. Fibril Aβs cause neuronal and synaptic loss, and extensive fibrils, along with activated microglia, accumulate to develop senile plaques. Fibril Aβs can also induce neurofibrillary tangles (NFTs) by activing various kinase and phosphatases, with neuronal loss and AD as a result. AChE, acetylcholinesterase; ApoE, apolipoprotein E; PS, presenilin.
Figure. 1. Schematic diagram of the pathogenesis of Alzheimer's disease (AD). In AD, amyloid precursor protein (APP) is hydrolyzed by β-secretase and then γ-secretase into β-amyloid (Aβ) that aggregates to form fibril Aβs. Fibril Aβs cause neuronal and synaptic loss, and extensive fibrils, along with activated microglia, accumulate to develop senile plaques. Fibril Aβs can also induce neurofibrillary tangles (NFTs) by activing various kinase and phosphatases, with neuronal loss and AD as a result. AChE, acetylcholinesterase; ApoE, apolipoprotein E; PS, presenilin.
Creative Bioarray provides you with APP transgenic models including but not limitted to:
- APPSL transgenic mouse model
- ApoB-100 transgenic mouse model
- TBA2.1 transgenic mouse model
- Tg4-42 (TBA83) mouse model
- SD- Apptm1sage
- Tg2576 mice model
Our Capabilities
- We offer APP transgenic models to screen drugs for treatment of Alzheimer's disease.
- We provide comprehensive behavioral and cognitive testing on AD model and the drug screening.
- We utilize LTP (long-term potentiation) assay to evaluate your preclinical drug candidates against the synaptic impairments in animals.
- We evaluate anti-oxidative stress in the brain of animals treated with drug candidates.
- We evaluate various biomarkers through WB, IHC, ELISA, sequencing, etc.
Assays available
- Learning and memory deficits tests
- Synaptic impairment
- Oxidative stress
- Neuroinflammation
- Glycogen synthase kinase-3 beta (GSK3β)
- Neuronal loss
- β-AP level
- Plaque load
- β-sheet load
- pE(3)-Aβ load
- Enzyme activity related to cholinergic system
- NMDA receptor function and excitotoxicity
- Mitochondrial dysfunction
- Brain slice staining and synaptic electrophysiology
With extensive experience in the field of AD, we are confident to help you to overcome any upcoming challenges. Our experts are fully capable of customizing our protocols and assays to meet your specific needs. With our help, we wish to facilitate your research with high efficiency.
Study examples
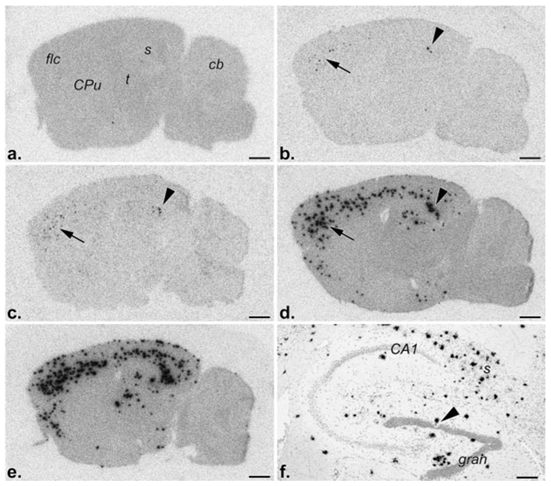 Figure 2 . Fibrillar Aβ deposits, i.e., diffuse and mature plaques, revealed by in vitro binding of monomeric 125I-Aβ1-40 to parasagittal brain sections followed by film radioautography. a, Nontransgenic control; 5 (b)-, 9 (c)-, 13 (d)-, and 17 (e)-month old PS2APP transgenic mice, respectively. Note the absence of deposits, i.e., binding, in the control, rare deposits in the subiculum (arrowhead) and frontolateral cortex (arrow) of a 5-month-old mouse, and their gradual, age-related increase in number and distribution at subsequent time points. f, Emulsion radioautography of a radiolabeled brain section of a 17-month-old mouse revealing the cellular distribution of Aβ deposits (black silver grains) in the hippocampal formation. Note, inter alia, the markedly reduced number of granule cells (arrowhead) in the vicinity of one cluster of silver grains, i.e., plaque. CA1, Ammon’s horn layer 1; cb, cerebellum; CPu, caudate putamen; gran, dentate gyrus granule cell layer; flc, frontolateral cortex; s, subiculum; t, thalamus. Scale bars:a–e, 1 mm; f, 200 µm.
Figure 2 . Fibrillar Aβ deposits, i.e., diffuse and mature plaques, revealed by in vitro binding of monomeric 125I-Aβ1-40 to parasagittal brain sections followed by film radioautography. a, Nontransgenic control; 5 (b)-, 9 (c)-, 13 (d)-, and 17 (e)-month old PS2APP transgenic mice, respectively. Note the absence of deposits, i.e., binding, in the control, rare deposits in the subiculum (arrowhead) and frontolateral cortex (arrow) of a 5-month-old mouse, and their gradual, age-related increase in number and distribution at subsequent time points. f, Emulsion radioautography of a radiolabeled brain section of a 17-month-old mouse revealing the cellular distribution of Aβ deposits (black silver grains) in the hippocampal formation. Note, inter alia, the markedly reduced number of granule cells (arrowhead) in the vicinity of one cluster of silver grains, i.e., plaque. CA1, Ammon’s horn layer 1; cb, cerebellum; CPu, caudate putamen; gran, dentate gyrus granule cell layer; flc, frontolateral cortex; s, subiculum; t, thalamus. Scale bars:a–e, 1 mm; f, 200 µm.
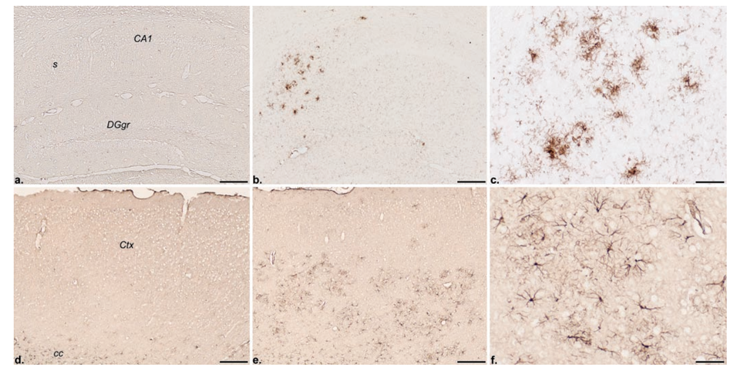 Figure 3 . Immunohistochemical localization of the inflammatory response markers CD45 (a–c) and GFAP (d–f ) in the hippocampal formation and frontolateral cortex, respectively, of a nontransgenic control (a, d) and a 13-month-old PS2APP transgenic mouse (b, c, e, f ). Note the absence of CD45 and GFAP immunoreactivities, i.e., microglioses and astroglioses, respectively, in the control but their discrete localization in the transgenic brain. CA1, Ammon’s horn layer 1; cc, corpus callosum; Ctx, neocortex; DGgr, dentate gyrus granular layer; s, subiculum. Scale bars: a, b, d, e, 200 µm; c, f, 50 µm.
Figure 3 . Immunohistochemical localization of the inflammatory response markers CD45 (a–c) and GFAP (d–f ) in the hippocampal formation and frontolateral cortex, respectively, of a nontransgenic control (a, d) and a 13-month-old PS2APP transgenic mouse (b, c, e, f ). Note the absence of CD45 and GFAP immunoreactivities, i.e., microglioses and astroglioses, respectively, in the control but their discrete localization in the transgenic brain. CA1, Ammon’s horn layer 1; cc, corpus callosum; Ctx, neocortex; DGgr, dentate gyrus granular layer; s, subiculum. Scale bars: a, b, d, e, 200 µm; c, f, 50 µm.
Quotation and ordering
If you have any special needs or questions regarding our services, please feel free to contact us. We look forward to cooperating with you in the future.
References
- Li X et al. Experimental models of Alzheimer's disease for deciphering the pathogenesis and therapeutic screening (Review). International Journal of Molecular Medicine. 2015; 37: 271.
- Grayson R et al. PS2APP transgenic mice, coexpressing hPS2mut and hAPPswe, show age-related cognitive deficits associated with discrete brain amyloid deposition and inflammation. Journal of Neuroscience the Official Journal of the Society for Neuroscience. 2003; 23: 8989.