Porcine Kidney Fibroblasts
Cat.No.: CSC-C4891L
Species: Pig
Source: Kidney
Cell Type: Fibroblast
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Never can cryopreserved cells be kept at -20 °C.
Porcine Kidney Fibroblasts (PKF) are primary cells taken from pig kidneys. Specifically, they originate from the interstitial connective tissue within the kidney. Morphologically, they are spindle-shaped elongated cells. PKFs have also been shown to possess prodigious synthesis and remodeling capabilities of ECM components. Functionally, PKFs are the cells responsible for creating the structural infrastructure of the kidney and thus play a role in tissue homeostasis as well as wound healing repair within the kidney. Additionally, PKFs in vitro have also been shown to proliferate well and adhere well to their substrate. Because of this, PKFs have been used extensively and have proven to be a robust model for porcine kidney cell behavior and function.
PKFs are especially useful when screening for drugs that aid in combating renal fibrosis. In response to pro-inflammatory cytokines or growth factors (i.e. TGF-β), fibroblasts transdifferentiate into myofibroblasts which deposit excessive amounts of collagen resulting in scarring of the tissue. For this reason, PKFs can be used to screen drugs that prevent fibrosis as well as identify pathways involved in chronic kidney disease. As pigs share many similar traits to humans, PKFs can be used to study cellular interactions with humans in xenotransplantation and immune rejection. In addition, PKFs are often used to propagate viruses and to produce biological reagents.
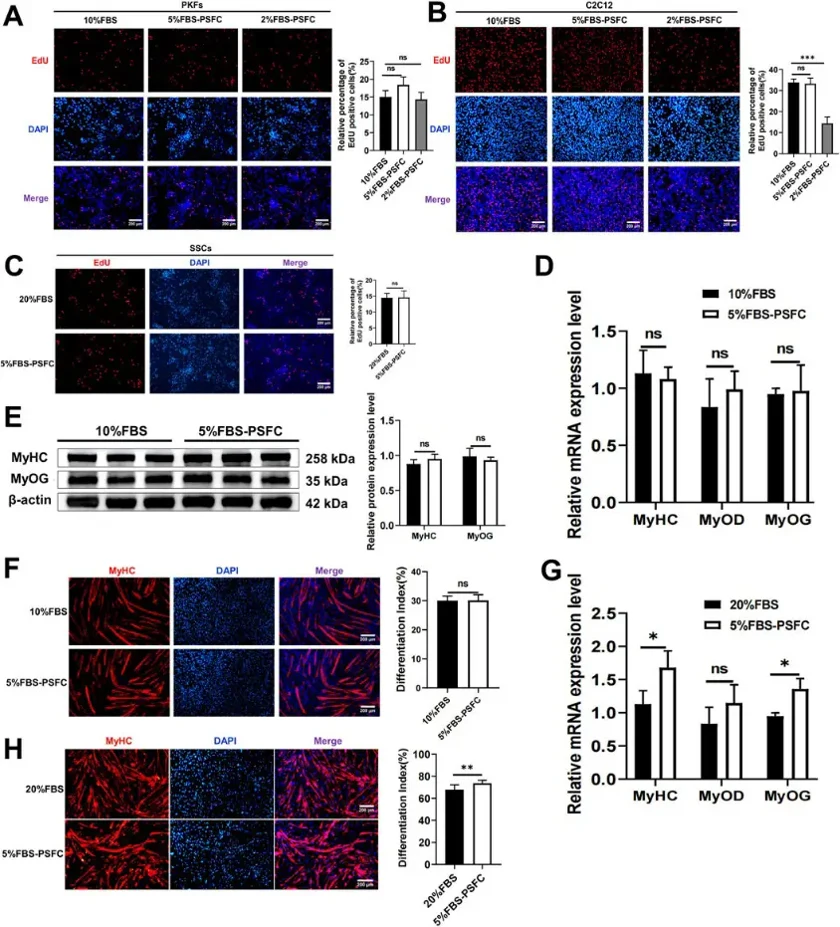
5% FBS-PSFC System Maintains Proliferation and Differentiation Capacities Across Diverse Cell Types
Low-serum culture systems enhance controllability, safety, and cost-effectiveness for tissue engineering, regenerative medicine, drug screening, and cultured meat production. LI's team developed a novel proliferation synergy factor cocktail (PSFC) under low-serum conditions to sustain cell proliferation and enhance transfection efficiency.
They validated their optimized PSFC culture platform across multiple cell types and species. Proliferation assays showed consistent capacity in porcine kidney fibroblasts (PKFs), myoblasts (C2C12), and mouse skeletal muscle satellite cells (SSCs) (Fig. 1A-C). All cell types had similar proliferation rates in 5% FBS-PSFC compared to conventional conditions. PKFs and C2C12, typically cultured in 10% FBS, were also tested in 2% FBS-PSFC. PKFs showed no significant difference in EdU+ cells between 5% and 2% FBS treatments compared to 10% FBS controls (Fig. 1A). However, C2C12 had significantly fewer EdU+ cells in 2% FBS-PSFC (Fig. 1B), indicating insufficient proliferation support at this serum level. C2C12 myoblasts showed no significant changes in MyoD, MyoG, and MyHC mRNA expression in 5% FBS-PSFC (Fig. 1D), with unchanged MyHC protein expression and fusion indices (Fig. 1E, F). In contrast, SSCs cultured in 5% FBS-PSFC exhibited enhanced myogenic differentiation, with upregulated MyoG and MyHC mRNA and increased MyHC+ myotube formation (Fig. 1G, H). Overall, the 5% FBS system supports robust proliferation and differentiation across various cell types and species, reducing serum requirements by 50%.
Ignoring the culture medium, the main consideration is that the cells are in poor condition when frozen or that the action is too slow during resuscitation leading to cell death. Remember that the most important thing is to thaw fast, and shake the cryotube from time to time so that it passes through the most vulnerable temperature band (-5 to 0°C) as quickly as possible.
Ask a Question
Average Rating: 5.0 | 1 Scientist has reviewed this product
High Quality
The cell products enabled us to get accurate and reliable results from our experiments.
12 Aug 2023
Ease of use
After sales services
Value for money
Write your own review