Human Bladder Epithelial Cells
Cat.No.: CSC-C4871L
Species: Human
Source: Bladder
Cell Type: Epithelial Cell
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Never can cryopreserved cells be kept at -20 °C.
Epithelial cells from the bladder (urothelial cells) line the inner surface of the urinary bladder, forming a specialized and highly impermeable barrier that prevents urine‑borne toxins and pathogens from reaching the underlying tissue. In vivo, the urothelium consists of basal, intermediate and umbrella cells; the latter express uroplakin (UPK1A/B, UPK2, UPK3A/B) and tight‑junction proteins (ZO‑1, claudin‑4) which confer its tensile strength and impermeability. In vitro, researchers can select from a range of urothelial cell models, from primary normal cells isolated from healthy bladder tissue to immortalized and cancer‑derived lines. Primary urothelial cells are typically cultured in urothelial‑specific medium with 10% fetal bovine serum, and retain the native 3‑layer architecture and UPK expression profile, making them ideal for in vitro studies of urothelial barrier‑function and toxicology. Immortalized normal urothelial cells like SV‑HUC‑1 (SV40‑transformed) provide a more stable non‑tumorigenic platform for long‑term experiments. Common bladder‑cancer lines include RT4 (low‑grade papillary carcinoma, doubling time 66-80 h), T24 (high‑grade carcinoma with H‑ras mutation, doubling time ≈19 h), 5637 (moderately differentiated, cytokine‑secreting) and UM‑UC‑3 (high‑grade, hyper‑triploid). These models have been used to investigate a variety of research topics: urothelial infection (UPEC invasion and intracellular bacterial communities), drug‑screening and antimicrobial‑peptide efficacy, bladder‑cancer signaling pathways (Ras‑MAPK, PI3K/AKT), environmental toxicology (BPA, heavy metals), and tissue‑engineering approaches for bladder reconstruction. Investigators can therefore select the appropriate cell line and culture system to faithfully recapitulate aspects of urothelial physiology and pathology in vitro.
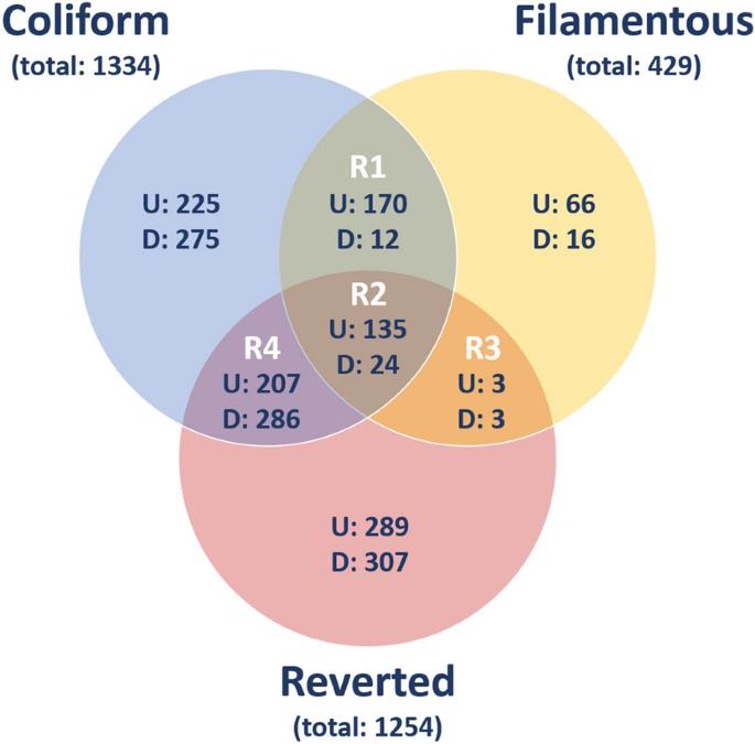
Transcriptional Alterations in Bladder Epithelial Cells in Response to Infection with Different Morphological States of Uropathogenic Escherichia coli
Uropathogenic Escherichia coli (UPEC) undergoes morphological changes that may enhance its ability to evade host responses and re-infect host cells. However, the impact of these changes on host activation and pathogenic potential is not well understood. Persson et al. found that the normal coliform induced the most significant gene alterations and canonical pathways, while the filamentous form had the least impact on host cell invasion.
HBEP cells were analyzed via mRNA microarray after bacteria transitioned from coli to filamentous forms and back. 2018 differentially expressed genes were identified, with at least a 2-fold change vs. controls. Each bacterial form induced distinct gene expression changes, visualized in a Venn diagram (Fig. 1). As a control, cells stimulated only with ceftibuten did not exhibit any significant changes unrelated to bacterial infection. In total, 341 genes were common between Coliform and Filamentous; 165 genes between Filamentous and Reverted, 652 genes between Coliform and Filamentous and 159 genes among all three stimulated groups are illustrated in Fig. 1. A group of genes with >100-fold upregulation includes IL17C, TNFAIP6 etc. These genes, except for ICAM1, were highly upregulated in cells stimulated with Coliform (Fig. 1).
Human Bladder Epithelial Cells from Creative Bioarray are isolated from normal human bladder tissue. The method we use to isolate primary epithelial cells was developed based on a combination of established and our proprietary methods. Prior to shipping, cells at passage 3 are detached from flasks and immediately cryo-preserved in vials.
Our Primary bladder epithelial cells have been tested for the expression of the following biomarkers by immunocytochemistry staining: CK5 (+), CK7 (+), CK8 (+), CK18 (+), P63(nuclear) (+), Adherens junction (AJ) protein E-cadherin (+), Tight junction (TJ) protein - Zona occludens 1(ZO-1) (+) and α-smooth muscle Actin (-).
These cells proliferate very slowly and may take up to 10-14 days to fully recover from cryopreservation. After passaging, the cells may take about 7-10 days to reach 70-90% confluence, depending on the seeding density.
It is recommended to use SuperCult® Complete Human Epithelial Cell Medium (cat# CM-1098X) for the culturing of Human Bladder Epithelial Cells.
Creative Bioarray ships frozen cells on dry ice. On receipt, immediately transfer frozen cells to liquid nitrogen (-180 °C) until ready for experimental use. Live cell shipment is also available on request.
Ask a Question
Average Rating: 5.0 | 1 Scientist has reviewed this product
Great products and excellent support
1) High-quality product 2) Very well organised and excellent support from product delivery and experiment design practicalities.
13 Nov 2022
Ease of use
After sales services
Value for money
Write your own review