Bovine Brain Microvascular Endothelial Cells
Cat.No.: CSC-C8069L
Species: Bovine
Source: Brain
Cell Type: Endothelial Cell; Microvascular Cell
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
The Bovine Brain Microvascular Endothelial Cell System consists of cryopreserved primary bovine brain microvascular endothelial cells from a pool of multiple donor brains, and basal medium for growth into confluent monolayers. The endothelial cells express cellular tight junctions (positive for the proteins ZO-1 and Claudin-1), and transport characteristics (P-Glycoprotein function) found in the blood brain barrier. Thus the system can be used in early stage ADME-Tox screening assays to demonstrate whether test compounds are likely to cross the blood brain barrier.
Bovine Brain Microvascular Endothelial Cells (BBMECs) are primary endothelial cells derived from microvasculature of the bovine brain. These cells have been extensively used as an in vitro model of the blood-brain barrier (BBB). They maintain several unique structural and functional characteristics associated with endothelial cells of the brain and are therefore considered physiologically relevant to studies of cerebrovascular biology, neuropharmacology, and drug delivery.
BBMECs grown in culture possess a cobblestone morphology and ability to form confluent tight monolayers with well-developed intercellular junctions. Characteristic endothelial markers, including CD31 (PECAM-1) and von Willebrand factor (vWF), as well as tight junction proteins such as claudin-5, occludin, and ZO-1 are expressed by BBMECs. Active transport mechanisms are also present such as P-glycoprotein and other efflux transporters. Overall, these properties allow BBMECs to serve as a model for both passive and active transport components of the BBB.
BBMECs have been utilized in many studies including: BBB physiology, neuroinflammation, neurotoxicity, CNS drug permeability, and drug transport. Due to their barrier integrity, physiologic properties and responses to various chemical and biological stimuli, BBMECs have been useful for translational research including pharmacology, toxicology, and disease pathology of neurological disorders.
Bioengineered 3D microvessels and complementary animal models reveal mechanisms of Trypanosoma congolense sequestration
Trypanosoma congolense sequesters to the vascular endothelium in mammalian hosts, influencing disease severity, but the determinants and mediators of this process are unknown. Porqueddu et al. investigated the roles of flow and endothelial tropism in parasite sequestration using 3D bovine endothelial microvessel models.
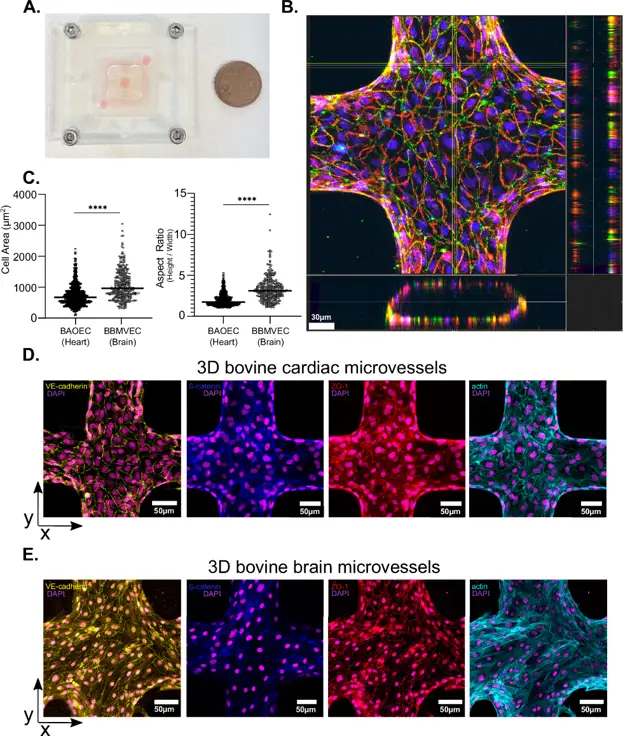
Previous research showed that different T. congolense strains accumulate in various organs in mouse models, causing distinct diseases. For example, the 1/148 strain accumulates in the brain, causing acute cerebral trypanosomiasis, while the IL3000 strain targets the heart, causing chronic disease. To study this organ preference in vitro, they created two microvessel systems: one using bovine aortic endothelial cells (BAOEC) to mimic the heart, and one using bovine brain microvascular endothelial cells (BBMVEC) to mimic the brain. Both cell types were confirmed as endothelial by immunostaining. BBMVEC had higher claudin-5 expression than BAOEC and expressed ICAM-1, VCAM-1, and EPCR. Both cell types were seeded in a 13 × 13 microfluidic network of 120 µm microvessels, mimicking post-capillary venules. After 3 days, they formed 3D monolayers with empty lumens (Fig. 1B). Immunofluorescence showed that BAOEC are smaller and rounder, while BBMVEC are larger and more elongated (Fig. 1C). Both expressed adherens (VE-cadherin, ß-catenin) and tight (ZO-1) junction markers (Fig. 1D and E). BAOEC had cortical actin, while BBMVEC had more stress fibres. In conclusion, they developed two in vitro microvessel systems to study organ-specific trypanosome sequestration.
Ask a Question
Write your own review
- You May Also Need