Rat Type Ⅱ Alveolar Epithelial Cells
Cat.No.: CSC-C5015S
Species: Rat
Source: Lung
Cell Type: Epithelial Cell
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Rat Type Ⅱ Alveolar Epithelial Cells (rT2AECs) from Creative Bioarray are isolated from the lung tissue of rats. The method we use to isolate renal collecting duct epthelial cells was developed based on a combination of established and our proprietary methods. The rPAAFs from Creative Bioarray are characterized by immunofluorescence with antibodies specific to pulmonary surfactant proteins (SP-A). Each vial contains 0.5x10^6 cells per ml and is delivered frozen.
Rat Type II Alveolar Epithelial Cells, also known simply as rat ATII cells or type II alveolar cells, are epithelial cells derived from rat lung alveoli. Rat ATII cells are one of two types of epithelial cells that make up the pulmonary alveolar epithelium and have several critical functions that contribute to normal lung physiology and alveolar homeostasis. Unlike type I alveolar epithelial cells which are involved in mediating gas exchange across the alveolar-capillary barrier, type II alveolar epithelial cells synthesize, secrete, and recycle pulmonary surfactant.
Rat type II alveolar epithelial cells are often cuboidal shaped and contain lamellar bodies. Lamellar bodies contain components of pulmonary surfactant such as phospholipids and surfactant proteins. These cells commonly express epithelial markers and surfactant-associated proteins such as surfactant protein A (SP-A), surfactant protein B (SP-B), surfactant protein C (SP-C), and epithelial cytokeratins when cultured in vitro and are used for identification purposes. Rat type II alveolar epithelial cells also act as progenitor cells for the alveolar epithelium. When the alveolar epithelium is damaged, they can proliferate and differentiate into type I alveolar epithelial cells to contribute to alveolar repair. They are also able to produce cytokines, chemokines, and antimicrobial peptides that contribute to the pulmonary immune response. Due to their ability to represent many critical processes in lung physiology in vitro, rat type II alveolar epithelial cells are frequently used to model many different aspects of lung development and disease. These cells have been used in studies examining pulmonary development, inflammation, acute lung injury, pulmonary fibrosis, lung infection, and drug toxicity.
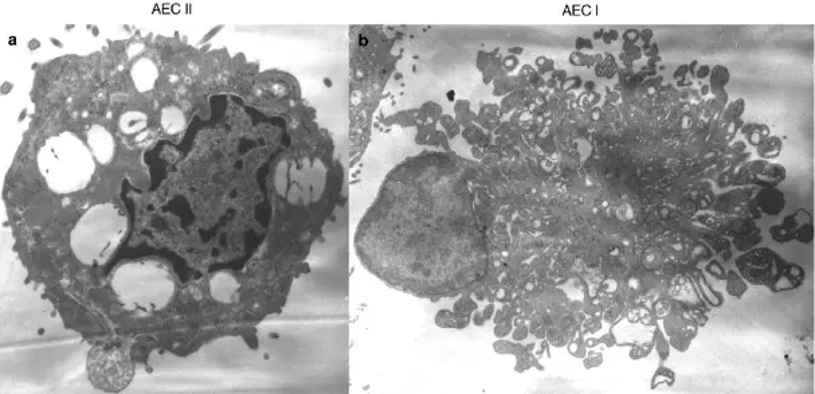
In Vitro Protective Effect of SH on NiO-NPs Induced Lung Injury
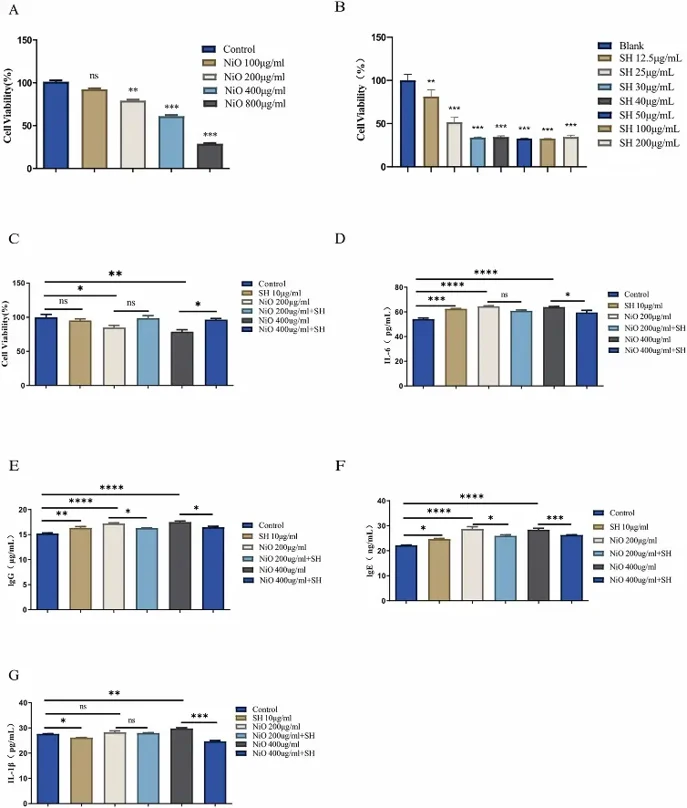
Nickel oxide nanoparticles (NiO-NPs) have been associated with inflammatory diseases. However, there is currently no specific treatment for NiO-NP-induced pneumonia. In the present study, cytotoxicity models were first established with rat type II alveolar epithelial cells (ACE-II).
Treatment with NiO-NPs caused concentration-dependent cytotoxicity. Concentrations of 100 μg/mL NiO-NPs caused no significant cytotoxicity; however, viability was significantly decreased at 200-800 μg/mL NiO-NPs (Fig. 1A). Thereafter, 400 μg/mL (86.37% viable) was chosen as the optimal dose for our in vitro experiments. Treatment with SH exhibited cytotoxicity at concentrations of 12.5 μg/mL (81.47% viability) and above. A concentration of 10 μg/mL was chosen for subsequent experiments (Fig. 1B). Furthermore, treatment with SH was not cytotoxic relative to the control group (Fig. 1C). SH treatment was unable to alleviate injury at 200 μg/mL NiO-NPs; however, SH treatment significantly attenuated viability at 400 μg/mL NiO-NPs. SH protects against NiO-NP-induced lung injury at certain concentrations without inducing cytotoxicity to cells.
Ask a Question
Write your own review
- You May Also Need