- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Rat Theca Cells from Creative Bioarray are isolated from the rat ovarian tissue. The method we use to isolate Rat Theca Cells was developed based on a combination of established and our proprietary methods. The Rat Theca Cells are characterized by immunofluorescence with antibodies specific to 3β-Hydroxysteroid dehydrogenase (3β-HSD). Each vial contains 0.5x10^6 cells per ml and is delivered frozen.
Rat theca cells, the androgen-producing stromal cells surrounding ovarian follicles, are a cornerstone model for reproductive endocrinology. Their advantages lie in: 1) Physiological fidelity-primary rat theca interna cells retain in vivo steroidogenic capacity (e.g., LH-stimulated androstenedione synthesis via CYP17A1), faithfully recapitulating follicular theca compartment function; 2) Pathophysiological relevance-they are pivotal for dissecting mechanisms of hyperandrogenism in polycystic ovary syndrome (PCOS), including theca cell hyperplasia and dysregulated steroidogenesis; 3) Experimental robustness-established isolation protocols yield high-viability cells amenable to siRNA, hormonal challenge, and metabolomic studies of cholesterol utilization; and 4) Translational utility-insights into theca-derived paracrine signals and enzyme regulation directly inform drug development for androgen excess disorders and ovulation dysfunction. Their defined gonadotropin responsiveness positions them as an irreplaceable platform for ovarian biology research.
Jiao-Tai-Wan Attenuate PCOS by Regulating Mitochondrial Cholesterol Import Through Suppression of SIRT1 Ubiquitination
Polycystic ovary syndrome (PCOS) has emerged as a common endocrine disorder that impacts female reproductive health. The traditional herbal formulation, Jiao-tai-wan (JTW), exhibits multiple biological activities, including improving insulin resistance, reducing oxidative stress, and mitigating inflammation. The clinical efficacy of JTW against PCOS has been reported, however, mechanistic studies remain absent.
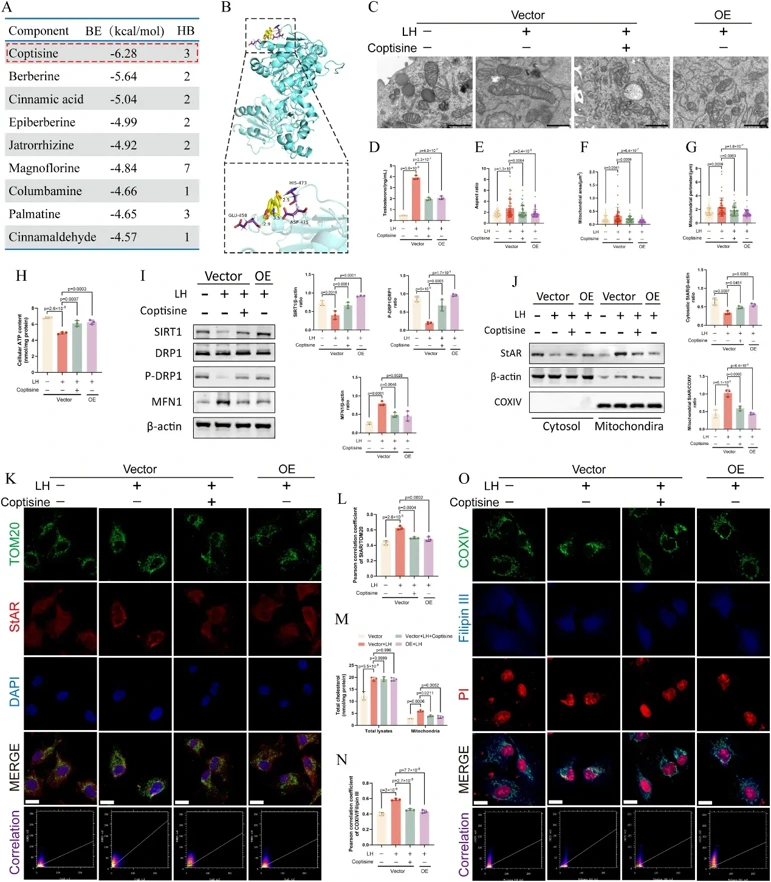
The PCOS rat model was established by injection of dehydroepiandrosterone (DHEA). The in vitro study utilized primary theca cells. Key pathways were identified via RNA sequencing, and the primary constituents of JTW were identified using UPLC fingerprinting. Further mechanistic investigations were conducted using techniques including network pharmacology, cell transfection, transmission electron microscopy imaging, confocal imaging, co-immunoprecipitation, CETSA, and SPR.
JTW attenuated abnormal ovulation, sex hormone imbalance, glycolipid metabolism disorders , and oxidative stress in PCOS rats. RNA sequencing revealed that JTW regulated the ovarian steroidogenesis pathway. Furthermore, JTW regulated mitochondrial dynamics and inhibited StAR localization to the outer mitochondrial membrane in the ovarian theca cells. SIRT1 was identified as the key target of JTW. Coptisine, a component of JTW, reversed abnormal mitochondrial dynamics in theca cells by upregulating SIRT1 expression, which in turn suppressed mitochondrial cholesterol import, thereby alleviating aberrant steroidogenesis. Coptisine intervention produced effects similar to SIRT1 overexpression, but SIRT1 knockdown blocked these effects. Notably, coptisine did not alter SIRT1 mRNA levels but enhanced SIRT1 protein expression by suppressing ubiquitination-mediated degradation. Coptisine weakened the interaction between the E3 ubiquitin ligase SMURF2 and SIRT1.
Ask a Question
Write your own review