Rat Tendon Fibroblasts
Cat.No.: CSC-C5131S
Species: Rat
Source: Tendon
Cell Type: Tenocyte
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Rat Tendon Fibroblasts from Creative Bioarray are isolated from the rat achilles tendon tissue. The method we use to isolate Rat Tendon Fibroblasts was developed based on a combination of established and our proprietary methods. The Rat Tendon Fibroblasts are characterized by immunofluorescence with antibodies specific to vimentin or fibronectin. Each vial contains 0.5x10^6 cells per ml and is delivered frozen.
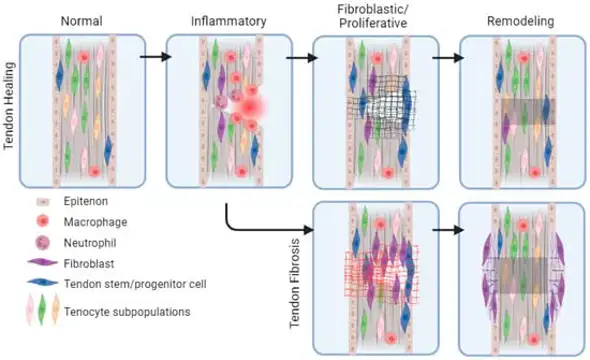
Rat Tendon Fibroblasts (RTFs), also called tenocytes when mature, are the main resident cell type in tendon tissues, including the Achilles tendon or tail tendon. They are specialized mesenchymal cells that synthesize, maintain and remodel the extracellular matrix (ECM), which is mostly made of Type I collagen fibres. Morphologically, RTFs exhibit an elongated spindle shape and are identified through expression markers such as vimentin, scleraxis (Scx), and tenomodulin (Tnmd) when isolated and cultured in vitro.
RTFs are commonly used to study mechanobiology due to their susceptibility to mechanical loading and tension. It has been shown that RTFs are used to study molecular mechanisms of tendinopathy and scarring, in which tenocytes switch from quiescent phenotype into a proliferative, fibroblast-like "synthetic" phenotype to initiate tissue repair. They have also been used to study the effect of growth factors like bFGF and TGF-β on collagen expression.
Due to tendons having limited vascularization and limited ability to regenerate, RTFs are used for tissue engineering or drug testing to mimic the tendon environment in vitro. This allows researchers to study how biochemical signaling and mechanical loading work in tandem to support musculoskeletal homeostasis or contribute to chronic inflammatory diseases.
PGEM Exhibited Excellent Performances in Cytocompatibility
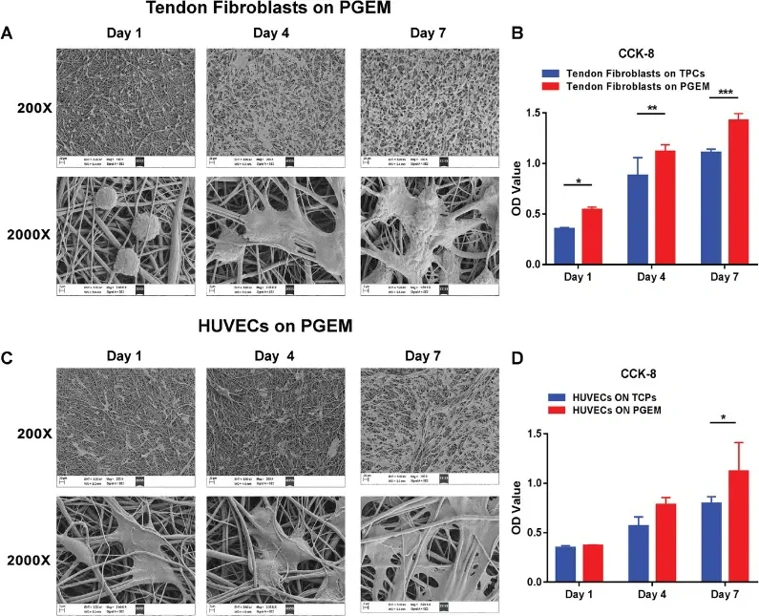
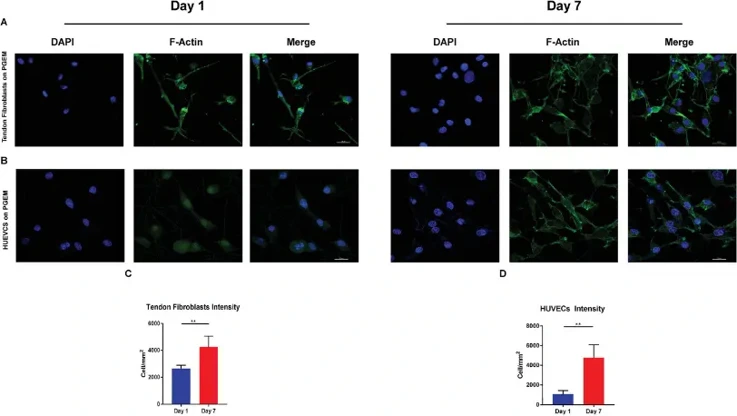
CMRCTs are challenging due to poor tendon regeneration and high retear rates. Here, Liu's team fabricated PLLA/gelatin electrospun membranes (PGEM) using electrospinning technology. They tested their Fourier transform infrared spectra and static contact angles, and evaluated cytocompatibility with rat tendon fibroblasts and human umbilical endothelial cells (HUEVCs).
Scanning electron microscopy (SEM) images (Fig. 1A, C) showed that PGEM provided an optimal growth environment for rat tendon fibroblasts and HUEVCs. On day 1, tendon fibroblasts appeared spherical. By day 4, both cell types exhibited polygonal shapes with emerging pseudopodia, indicating good initial adhesion and attachment to the nanofibers. On day 7, cellular fusions were observed, with cells spreading out and adhering tightly to the nanofibers via pseudopodia, which also suggested enhanced migration. Thus, PGEM effectively promoted cell adhesion and migration, demonstrating good cytocompatibility. CCK-8 assay results (Fig. 1B, D) indicated that PGEM enhanced the proliferation of rat tendon fibroblasts and HUEVCs. The optical density (OD) values increased over the culturing period. For HUVECs, proliferation on PGEM was comparable to that on tissue culture plates (TCPs) (Fig. 1B). Notably, tendon fibroblasts on PGEM had higher OD values than those on TCPs at each time point (Fig. 1D), suggesting faster proliferation on PGEM.
Ask a Question
Write your own review