Rat DRG Neurons
Cat.No.: CSC-C5138S
Species: Rat
Source: Spinal Cord
Cell Type: Neuron
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Rat DRG Neurons from Creative Bioarray are isolated from the rat spinal cord tissue. The method we use to isolate Rat DRG Neurons was developed based on a combination of established and our proprietary methods. The Rat DRG Neurons are characterized by immunofluorescence with antibodies specific to neuron specific enolase (NSE). Each vial contains 0.5x10^6 cells per ml and is delivered frozen.
Rat DRG (Dorsal Root Ganglion) Neurons are primary sensory neurons that are acutely dissociated from the dorsal root ganglia of rats. They are a widely used cell model for studying the peripheral sensory nervous system, which is responsible for sensing and transmitting information from the skin, muscles, joints, and internal organs to the spinal cord and brain. Rat DRG neurons represent different sensory modalities, such as nociceptive (pain), mechanosensory (touch, pressure), and thermosensory (temperature) neurons, making them an attractive in vitro system for studying sensory transduction and excitability. Additionally, since the neurons are directly isolated from DRG and are known to express specific sensory-related receptors and ion channels, they are well suited for mechanistic studies to understand action potential generation, calcium signaling, and synaptic communication in peripheral neurons.
Cultured Rat DRG neurons preserve their physiological and morphological attributes which include diverse neuronal subpopulations representing various sensory phenotypes, polarized morphology and neurite extension capabilities along with the expression of crucial sensory-related ion channels and receptors like voltage-gated sodium and calcium channels and TRP channels. Therefore, they are a valuable model system for investigating the mechanisms underlying pain, including inflammatory pain, neuropathic pain, and chemotherapy-induced peripheral neuropathy. They are also used for studying axonal injury and regeneration, neuroinflammation, and neuron-glia interactions in the peripheral nervous system. In drug discovery, Rat DRG neurons are commonly used to assess the efficacy and safety of analgesic compounds, ion channel modulators, and neuroprotective agents.
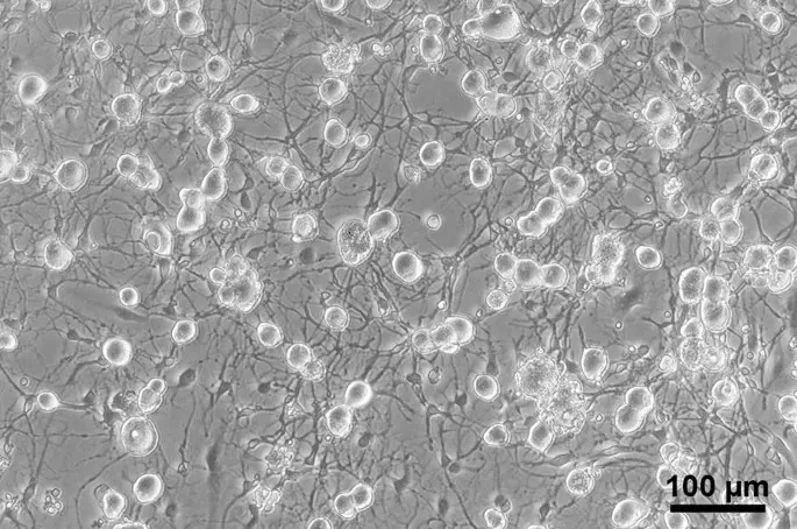
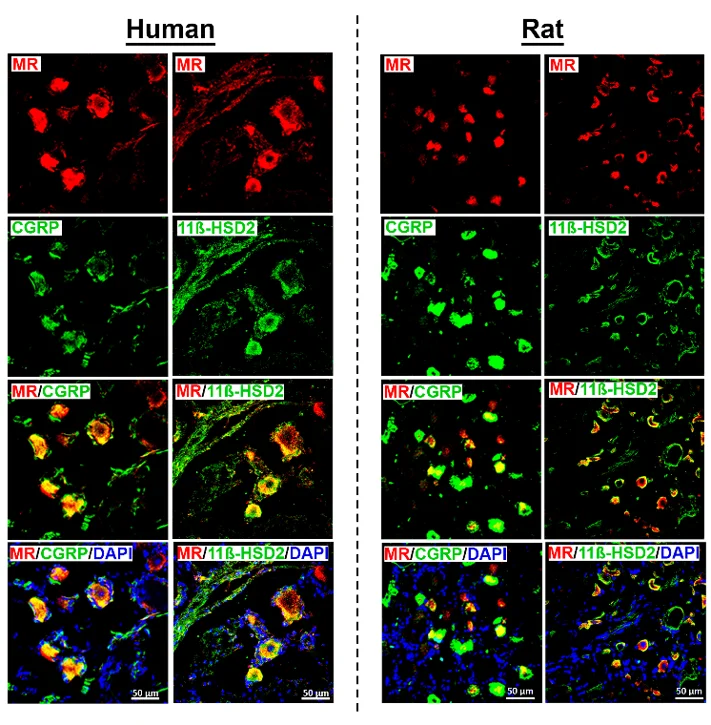
MR Receptor Detection in CGRP-IR Sensory Neurons Colocalizing with Its Protecting Enzyme 11ß-HSD2 in Human and Rat DRG Neurons
Activation of mineralocorticoid receptor (MR) is classically in the kidney. However, MR is also expressed in other extrarenal tissues including the heart, brain and dorsal root ganglion (DRG) neurons. Mousa et al. examined the expression and colocalization of MR, 11β-HSD2, aldosterone, and CYP11B2 in human DRG neurons and expression of pain associated signaling molecules to identify as potential targets of genomic regulation of pain.
They detected MR immunoreactivity in DRG neurons from both human and rat using specific antibodies. Rat DRG neurons appeared smaller than human ones under the same magnification. MR immunoreactivity was mainly found in a subpopulation of peripheral CGRP-IR sensory neurons, but not all CGRP-IR neurons had MR immunoreactivity, and some MR-IR neurons did not express CGRP. In human DRG neurons, MR-IR was mostly seen in small- to medium-diameter neurons (25-60 µm), consistent with a nociceptive phenotype, while it was less frequent in large-diameter neurons (≥70 µm). Double immunofluorescence confocal microscopy showed colocalization of MR with 11ß-HSD2 in both human and rat DRG. Quantification of human DRG tissue samples (n = 13-20) indicated that about 75% of MR-IR neurons were also CGRP-IR, and 43% were 11ß-HSD2-IR, suggesting partial protection of MR from circulating glucocorticoids.
Ask a Question
Write your own review