Rat Dendritic Cells
Cat.No.: CSC-C5063S
Species: Rat
Source: Bone Marrow
Cell Type: Dendritic Cell
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Rat Dendritic cells (rDCs) from Creative Bioarray are isolated from the rat bone marrow tissue. The method we use to isolate rDCs was developed based on a combination of established and our proprietary methods. The rDCs are characterized by immunofluorescence with antibodies specific to CD11c.
Rat dendritic cells are antigen-presenting cells (APCs) obtained from rats and used to study dendritic cell function. APCs are critical for the activation and regulation of adaptive immune responses. Dendritic cells (DCs) are professional antigen-presenting cells, capable of capturing antigens, processing them, and presenting antigen peptide/MHC complexes to naïve T lymphocytes. Dendritic cells link the innate and adaptive immune systems. Dendritic cells are collected from several tissues such as bone marrow, spleen, lymph nodes and peripheral blood in rats. Rat dendritic cells possess several immunologic properties that are analogous to dendritic cells found in other mammals.
They express cell surface molecules required for antigen presentation and T-cell activation, including major histocompatibility complex class II (MHC II), CD80, CD86 and CD11c. Immature dendritic cells will mature upon encountering pathogens/inflammatory signals. Dendritic cell maturation is defined by upregulation of co-stimulatory molecules, increased antigen-presenting capabilities, and migration to lymphoid organs to initiate T-cell activation. Depending on maturation signals, dendritic cells can promote strong immune responses or induce tolerance.
Typically rat dendritic cells are produced in vitro by culturing bone marrow-derived precursor cells with cytokines such as granulocyte-macrophage colony-stimulating factor (GM-CSF) and interleukin-4 (IL-4). They serve as an experimental tool that can be used to study antigen presentation, regulation of immunity, and mechanisms of dendritic cell maturation. Dendritic cells isolated from rats are used broadly in immunology, vaccine research, transplantation, autoimmune disease research, infection, and tumor immunity. Since rats are commonly used in pre-clinical models of disease, rat dendritic cells allow for examination of immune responses and testing of immunomodulatory therapeutics.
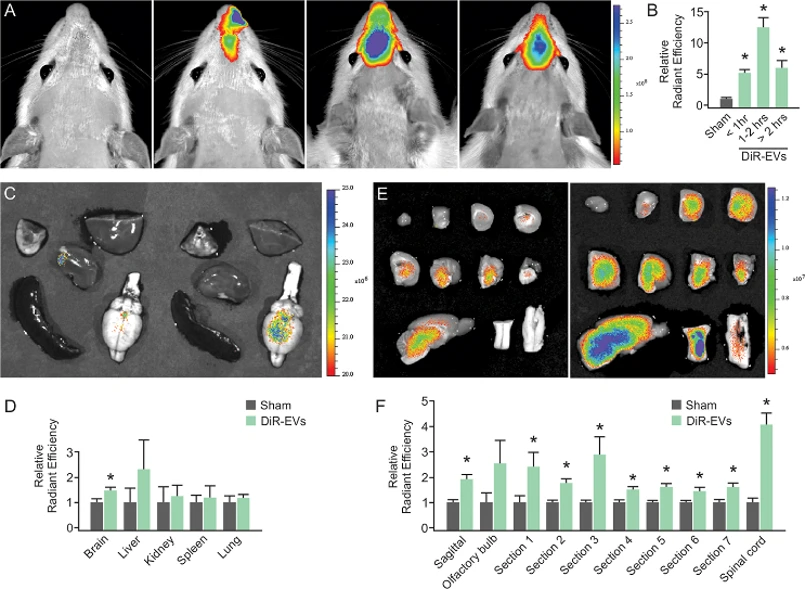
IFNγ-DC-Evs Distribute Throughout the Brain Following Nasal Administration
IFNγ-stimulated rat dendritic cell extracellular vesicles (IFNγ-DC-EVs) contain pro-myelinating miRNAs and preferentially enter oligodendrocytes. While functional studies suggested CNS entry, direct biodistribution evidence was lacking. Pusic et al. demonstrated brain-wide distribution and oligodendrocyte-specific uptake of nasally administered IFNγ-DC-EVs.
IFNγ-DC-EVs were labeled with Xenolight DiR (DiR-EVs) for IVIS imaging. Nasal administration resulted in significantly higher fluorescence in the rostral brain compared to sham controls, peaking at 1-2 hours post-administration (Fig 1A, B). Explanted organ imaging revealed significantly elevated brain fluorescence in DiR-EV treated animals, with no significant differences in liver, kidney, spleen, or lung (Fig 1C, D). Spatial distribution analysis using sagittal and coronal brain sections (Fig 1E) showed significantly increased fluorescence across all regions except the olfactory bulb (Fig 1G). Notably, the spinal cord showed the highest enrichment, demonstrating robust central nervous system penetration following nasal delivery.
Ask a Question
Write your own review