Mouse Internal Anal Sphincter Smooth Muscle Cells
Cat.No.: CSC-C5331S
Species: Mouse
Source: Anus
Cell Type: Smooth Muscle Cell
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Mouse internal anal sphincter smooth muscle cells from Creative Bioarray are isolated from the mouse internal anal sphincter tissue. The method we use to isolate mouse internal anal sphincter smooth muscle cells was developed based on a combination of established and our proprietary methods. The mouse internal anal sphincter smooth muscle cells are characterized by immunofluorescence with antibodies specific to α-SMA. Each vial contains 0.5x10^6 cells per ml and is delivered frozen.
Mouse Internal Anal Sphincter Smooth Muscle Cells (MIASMCs) are smooth muscle cells isolated from the internal anal sphincter (IAS) of mice. The IAS is a specialized band of circular smooth muscle that maintains basal anal tone and involuntary (autonomic) continence.
MIASMCs display classic elongated, fusiform morphology and proliferate as adherent monolayers. These cells express α-smooth muscle actin (α-SMA), smooth muscle myosin heavy chain (SM-MHC), calponin and SM22α, smooth muscle biomarkers that identify them as contractile cells. In appropriate conditions, they maintain phenotypic responsiveness to neurotransmitters and vasoactive agents such as nitric oxide (NO), acetylcholine, vasoactive intestinal peptide (VIP) and endothelin, which mediate IAS relaxation and contraction. These cells have been utilized to study the pathophysiology of fecal incontinence, hypertensive anal sphincter disorders, Hirschsprung disease and age-related changes. They allow for examination of intracellular calcium signaling, RhoA/ROCK activation, myosin phosphorylation and oxidative stress induced smooth muscle pathophysiology.
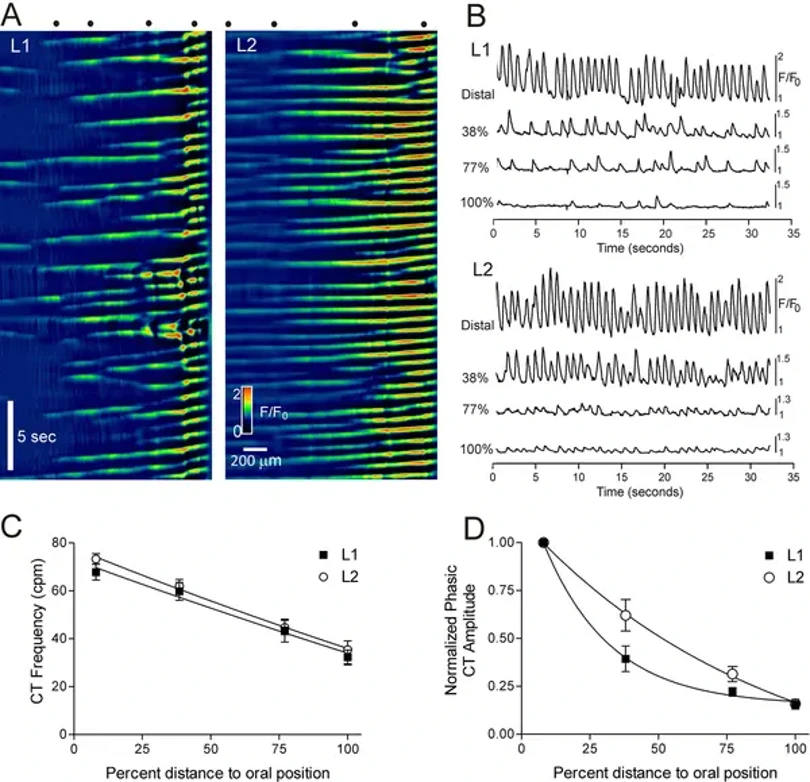
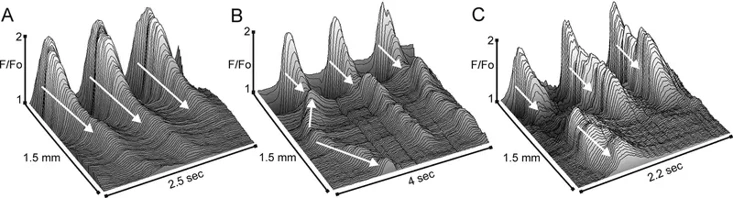
Spatiotemporal Properties of Ca2+ Transients in Smooth Muscle Cells of the Mouse Internal Anal Sphincter
The internal anal sphincter (IAS) generates slow waves (SWs) and tone via L-type Ca²⁺ channels (CavL), suggesting phasic activity contributes to tone generation. Cobine et al. examined spatiotemporal properties of Ca²⁺ transients (CTs) in IAS smooth muscle cells (SMCs) from 29 SM-GCaMP rectoanal tissues using confocal microscopy (4× magnification).
CTs consistently originated at the distal extremity and conducted proximally (Fig. 1A). Distal CT frequencies averaged 67.9±3.4 cpm (L1) and 73.3±2.2 cpm (L2), with no significant difference. Both frequency and amplitude declined proximally, with similar distance-dependent relationships in L1 and L2 muscles (Figs. 1C, D). While CTs consistently arose distally, conduction rates varied due to ectopic origins occurring at ~23% of distal frequency. These ectopic CTs conducted bidirectionally and occasionally collided with distal-originating CTs (Figs. 2B, C). CT conduction velocity was significantly faster in L2 versus L1 muscles (1.87±0.15 vs. 1.03±0.10 mm/sec; n=12, p<0.05), remaining significant after normalizing for muscle mass (1.31±0.10 vs. 1.03±0.10 mm/sec; n=12, p<0.05).
Ask a Question
Write your own review
- You May Also Need