Mouse Carotid Artery Smooth Muscle Cells
Cat.No.: CSC-C5356S
Species: Mouse
Source: Artery
Cell Type: Smooth Muscle Cell
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Mouse carotid artery smooth muscle cells from Creative Bioarray are isolated from the mouse carotid artery tissue. The method we use to isolate mouse carotid artery smooth muscle cells was developed based on a combination of established and our proprietary methods. The mouse carotid artery smooth muscle cells are characterized by immunofluorescence with antibodies specific to α-SMA. Each vial contains 0.5x10^6 cells per ml and is delivered frozen.
Mouse Carotid Artery Smooth Muscle Cells (MCASMCs) are primary vascular smooth muscle cells originally isolated from mouse carotid arteries. MCASMCs are commonly used as a model of vascular smooth muscle cells and arterial remodeling in vitro. MCASMCs have a typical spindle-shaped, elongated morphology and proliferate as adherent monolayers. They express canonical smooth muscle markers such as α-smooth muscle actin (α-SMA), smooth muscle myosin heavy chain (SM-MHC), calponin, and SM22α (Tagln). MCASMCs can phenotypically switch from contractile to synthetic/proliferative phenotype with different culture conditions and stimuli, which is consistent with characteristics of other smooth muscle cells found in vascular beds. MCASMCs are frequently utilized to study vascular injury and disease mechanisms in vitro.
MCASMCs have been used to study many vascular-related diseases such as atherosclerosis, restenosis due to angioplasty, hypertension, aneurysm, and vascular calcification. They have been used as an in vitro model to study cell proliferation and migration, extracellular matrix remodeling, oxidative stress, inflammatory signaling, and growth factor-mediated signaling pathways such as PDGF, TGF-β, MAPK, PI3K/AKT, NF-κB.
Macrophage-Derived Exo Facilitate SMC Cell Proliferation and Migration via BRCA1 Delivery
Yu's team investigated how macrophage-derived exosomes (Exo) delivering BRCA1 protein affect vascular smooth muscle cell (VSMC) function in vascular remodeling. In coronary artery disease (CAD), VSMCs transition from quiescent contractile to synthetic phenotypes with enhanced migration and proliferation, driving intimal thickening and disease progression.
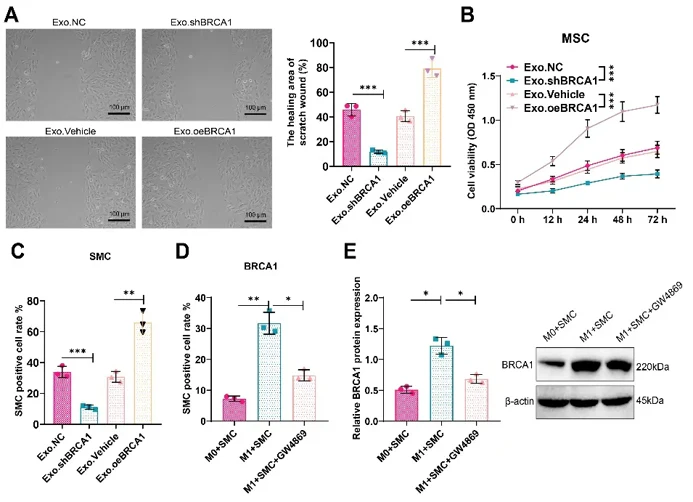
Using Exo with modulated BRCA1 expression, scratch assays showed Exo.shBRCA1 inhibited VSMC migration while Exo.oeBRCA1 promoted migration versus controls (Fig. 1A). CCK8 assays revealed corresponding effects on viability (Fig. 1B), and EdU proliferation assays confirmed Exo.shBRCA1 reduced while Exo.oeBRCA1 increased VSMC proliferation (Fig. 1C).
To validate direct BRCA1 transfer, they established an M1 macrophage-VSMC co-culture system. Ana-1 macrophages were polarized to M1 phenotype with LPS/IFN-γ, then co-cultured with VSMCs ± GW4869 (Exo release inhibitor). The M1+SMC group showed highest VSMC proliferation and BRCA1 protein expression, both markedly reduced by GW4869 (Fig. 1D, E), demonstrating that M1 macrophage-derived Exo directly transfer BRCA1 to VSMCs to regulate their function.
Ask a Question
Write your own review
- You May Also Need