Hamster Bone Marrow Macrophages
Cat.No.: CSC-C4758L
Species: Hamster
Source: Bone Marrow
Cell Type: Macrophage
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Never can cryopreserved cells be kept at -20 °C.
Hamster Bone Marrow Macrophages (hamBMDMs) are primary immune cells isolated from hamster bone marrow tissue, usually that of the Syrian (golden) hamster (Mesocricetus auratus). HamBMDMs serve as an in vitro primary cell model that can be used to study innate immune signaling, macrophage physiology, and host-pathogen interactions in hamster disease models.
HamBMDMs can be created by isolating progenitor cells from bone marrow and stimulating their differentiation into macrophages with macrophage colony-stimulating factor (M-CSF). Phenotypically, these cells are similar to macrophages that can be found in vivo, demonstrating a classical macrophage cell shape and many of the functional properties of macrophages, such as phagocytosis, cytokine and chemokine production, and response to inflammatory triggers such as LPS and interferons. HamBMMs can be polarized towards classically activated (M1-like) or alternatively activated (M2-like) macrophages using established cell culture conditions.
Hamster Bone Marrow Macrophages have been used to study infectious disease and immunology as the hamster is used as a translational animal model for many viruses, bacteria, and parasites, as well as diseases like those affecting the respiratory system and zoonotic infections. HamBMMs may have immune signaling pathways and cytokine production unique from that of mouse macrophages that better represents signaling in vivo.
Hamster Bone Marrow-Derived, Macrophage-Like Cells (Hambmdms) Express Macrophage Characteristics and Show High Phagocytic Capacity
The basis for differential susceptibility to Leishmania donovani infection remains poorly understood. To identify intrinsic cellular features contributing to host-specific outcomes, Jenkins et al. compared bone marrow-derived macrophages from susceptible hamsters (hamBMDMs) and resistant mice (mBMDMs).
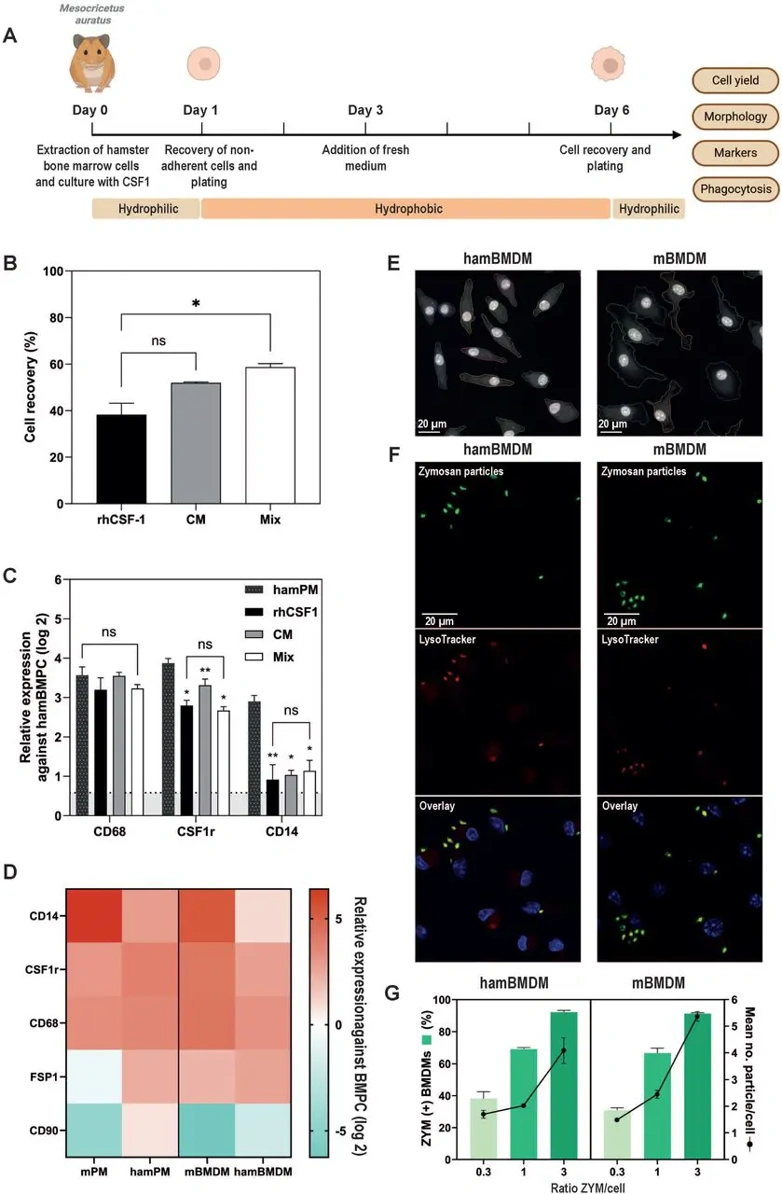
Large-scale production of quiescent macrophages requires CSF1-mediated differentiation of BMPCs rather than primary cell isolation, which alters activation states. Adapting mBMDM protocols to hamsters-critical for visceral leishmaniasis research-is hindered by lack of autologous CSF1. They therefore screened alternative CSF1 sources (Fig. 1A). E. coli-derived recombinant CSF1 yielded poor recovery, whereas HEK293-produced rhCSF1 (100 ng/ml) supported robust differentiation, indicating post-translational modifications enhance activity (Fig. 1B). BHK conditioned medium achieved comparable recovery, with 60% improvement using rhCSF1 plus 5% CM. Differentiation was assessed by RT-qPCR for CD68, CSF1r, and CD14 using hamBMPCs as calibrator and hamPMs as controls (Fig. 1C). CD68 and CSF1r reached hamPM levels under all conditions, while CD14 remained lower-a known macrophage subpopulation heterogeneity. Using BHK cells as calibrator revealed high baseline CD14 in hamBMPCs, explaining modest enrichment. They selected rhCSF1 alone to avoid CM confounders (Fig. 1C). Comparative profiling of hamBMDMs, hamPMs, mBMDMs, and mPMs confirmed similar macrophage marker expression between species, with minimal fibroblast contamination (FSP1, CD90; Fig. 1D). Both BMDM types exhibited characteristic morphology-flat, irregular bodies with round-to-oval nuclei (Fig. 1E)-comparable to human PBMC-derived macrophages. Functional validation using fluorescent zymosan particles demonstrated equivalent phagocytic capacity, with 92% of both hamBMDMs and mBMDMs internalizing particles at lowest concentration (Fig. 1F, G). These results validate rhCSF1-differentiated hamBMDMs as bona fide macrophages that faithfully reproduce mBMDM morphology, gene expression, and phagocytic function, providing a robust platform for comparative studies of Leishmania susceptibility.
Ask a Question
Write your own review
- You May Also Need