Canine Hepatocytes
Cat.No.: CSC-C4845L
Species: Dog
Source: Liver
Cell Type: Hepatocyte
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Never can cryopreserved cells be kept at -20 °C.
Canine Hepatocytes are liver parenchymal cells isolated from canine liver tissue that provide a relevant in vitro tool to study canine liver function, metabolism, and drug hepatotoxicity. Hepatocytes compose the majority of parenchymal cells in the liver and are responsible for most liver functions including xenobiotic metabolism, bile acid metabolism, protein synthesis, and energy homeostasis.
Maintained in culture, Canine Hepatocytes are polygonal in shape with visible cell-cell junctions. Additionally, these cells will form bile canaliculi-like structures when provided with optimal conditions. Canine Hepatocytes express various markers and metabolic enzymes characteristic of hepatocytes such as albumin, CYP isoforms, and drug transporter proteins. For these reasons, Canine Hepatocytes can be used to physiologically relevant assess metabolism and clearance of drugs and other chemicals. While primary hepatocytes can lose some liver-specific functions with prolonged culture time, they generally maintain metabolic functions within initial passages.
Due to their intrinsic role in drug metabolism, Canine Hepatocytes find frequent application in veterinary pharmacology, toxicology, and drug metabolism studies to predict species-specific metabolism as well as potential hepatotoxicity. Canine Hepatocytes can also be used to study canine liver pathologies, for drug candidate screening, and in translational science.
Kupffer Cells and Hepatocytes: a Key Relation in the Context of Canine Leishmaniasis
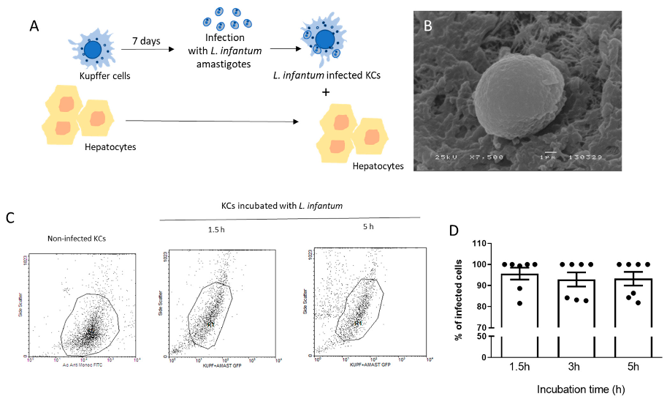
Canine visceral leishmaniasis (CanL), caused by Leishmania infantum, targets the liver. However, communication between hepatocytes and infected Kupffer cells (KCs), which may determine local immune responses during infection remains unknown. Rodrigues et al. generated a co-culture model of canine hepatocytes and L. infantum-infected autologous KCs.
To simulate natural liver infection, L. infantum amastigotes were in vitro differentiated from promastigotes by pH reduction and temperature increase, mimicking intracellular vacuole conditions and mammalian body temperature. Successful differentiation was confirmed by SEM as amastigotes displayed the rounded morphology and lack of flagellum typical of this life stage (Fig. 1B). Axenic amastigotes were able to infect primary KCs isolated from spleens (Fig. 2A) and did so successfully after 1.5 h of exposure. Infectivity assays were conducted up to 5 h post-exposure, beyond which KC lysis was too great (Fig. 1C, D). KCs were therefore infected for 5 h prior to establishment of co-culture with autologous hepatocytes (Fig. 2B, C) to recapitulate physiological infection of hepatocytes in vivo and allow interrogation of the mechanisms underlying this interaction (Fig. 1A).
Ask a Question
Write your own review
- You May Also Need