Rat Esophageal Smooth Muscle Cells
Cat.No.: CSC-C9366W
Species: Rat
Source: Esophagus
Morphology: Multipolar
Cell Type: Smooth Muscle Cell
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Rat Esophageal Smooth Muscle Cells are smooth muscle cells derived from the muscular layer of the esophagus of rats. The esophagus is a muscular organ that forms part of the gastrointestinal tract and functions to transport food from the pharynx to the stomach. This movement is accomplished by rhythmic contractions of smooth muscle called peristalsis. The layer of smooth muscle cells that make up the wall of the esophagus are responsible for generating the contractile activity that produces normal peristalsis. Rat esophageal smooth muscle cells are spindle-shaped cells that express smooth muscle markers such as α-smooth muscle actin (α-SMA), smooth muscle myosin heavy chain (SM-MHC), calponin, and SM22α.
Like other smooth muscle tissue from the gastrointestinal tract, rat esophageal smooth muscle cells share many of the same physiological properties as in vivo smooth muscle. This includes contractile responses to neurotransmitters, hormones, and mechanical stimuli that modulate muscle contraction and relaxation. Rat esophageal smooth muscle cells are often used to study gastrointestinal motility, smooth muscle contraction, and signaling mechanisms that regulate smooth muscle. Additionally, they can be used to study disease states such as motility disorders, inflammatory damage to smooth muscle, and tissue remodeling. Rats are frequently used as animal models of disease and are a common model for studying the gastrointestinal system. For this reason, rat esophageal smooth muscle cells can be used as an experimental tool to study mechanisms involved in esophageal function. Rat esophageal smooth muscle cells have been used to study neuromuscular regulation, receptor function, pharmacological effects, and therapeutic compounds on gastrointestinal smooth muscle.
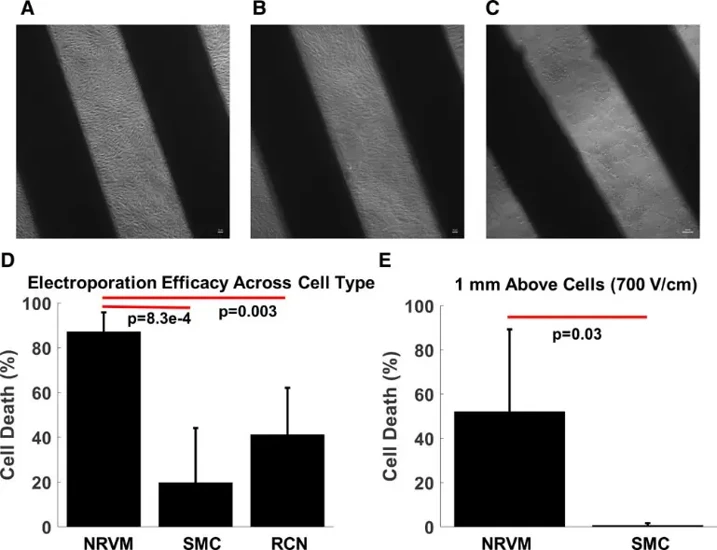
In Vitro Cell Selectivity of Reversible and Irreversible: Electroporation in Cardiac Tissue
Pulmonary vein isolation treats atrial fibrillation but risks incomplete lesions, reconnection, and collateral damage to the phrenic nerve and esophagus. Electroporation offers potential for permanent isolation with cardiac selectivity, but evidence for selective myocardial injury is lacking. Hunter et al. investigated whether electroporation achieves selective cell-type-specific ablation.
Monolayers of neonatal rat ventricular cardiomyocytes, rat cortical neurons, and esophageal smooth muscle cells were stained with propidium iodide to measure shock-induced cell death. Biphasic shocks (10 ms) were delivered from line electrodes (1 mm separation), and cardiomyocytes were optically mapped for conduction. Conduction block required 400±50 V/cm with electrode contact versus 690±70 V/cm at 1 mm distance (P<0.01). At 400 V/cm with contact, cardiomyocytes showed highest cell death (~60%), neurons intermediate (~40%), and smooth muscle cells lowest (~20%) (Fig. 1). At 1 mm distance, smooth muscle cells were nearly unaffected. These findings demonstrate that electroporation achieves intrinsic cell-type selectivity without confounding clinical factors, though electrode proximity remains critical for efficacy, supporting electroporation as a more selective modality for pulmonary vein isolation.
Ask a Question
Write your own review