Rat Bladder Epithelial Cells
Cat.No.: CSC-C9282J
Species: Rat
Source: Bladder
Cell Type: Epithelial Cell
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Rat Bladder Epithelial Cells (RBEC) are epithelial cells collected from mucosal lining of rat urinary bladder. The urinary bladder epithelium, also known as urothelium is a specialized stratified epithelium that lines the interior of the bladder. Bladder epithelium provides a barrier between urine and underlying tissue. Bladder epithelial cells consist of basal cells, intermediate cells and highly differentiated apical umbrella cells which function to maintain permeability barrier of bladder and protect underlying tissue from toxic components of urine. Rat bladder epithelial cells express many of the same physiological and molecular characteristics as native bladder epithelial cells including expression of epithelial markers cytokeratins, tight junction proteins, and proteins involved in barrier function like uroplakins.
In culture, rat bladder epithelial cells serve as an excellent model for urinary tract physiology, epithelial barrier function, and epithelial response to mechanical or chemical insult. Rat bladder epithelial cells are commonly used to study bladder inflammation, infection and epithelial injury. Rat bladder epithelial cells have also been used to study urothelial regeneration and differentiation. Rat bladder epithelial cells are also commonly used as experimental system for testing drug cytotoxicity and permeability of drugs, environmental toxicants, or biomaterials that may impact the urinary tract. Rat bladder epithelial cells are commonly used in the study of mammalian urinary tract disorders including interstitial cystitis, mechanisms of bladder cancer and urinary tract infections due to ease of correlation with rat disease models.
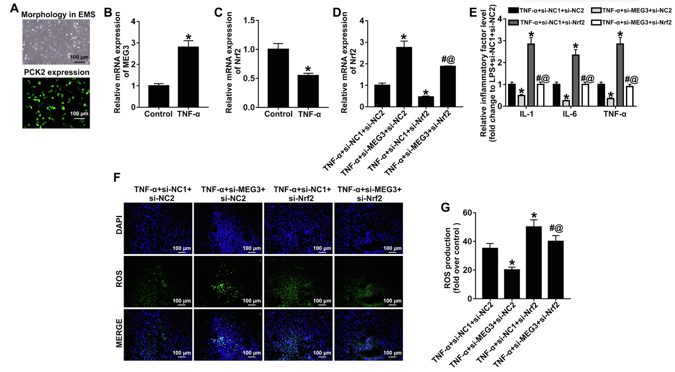
Downregulation of MEG3 Alleviates TNF-α-induced Inflammation, Oxidative Stress and Apoptosis in Rat Bladder Epithelial Cells through Upregulation of Nrf2.
Interstitial cystitis (IC), a chronic pain syndrome characterized by urinary frequency, urgency, and bladder or pelvic floor pain, severely affects the quality of life of patients. Wang's team investigated the role and mechanism of long noncoding RNA Maternally Expressed Gene3 (lncRNA MEG3) in IC.
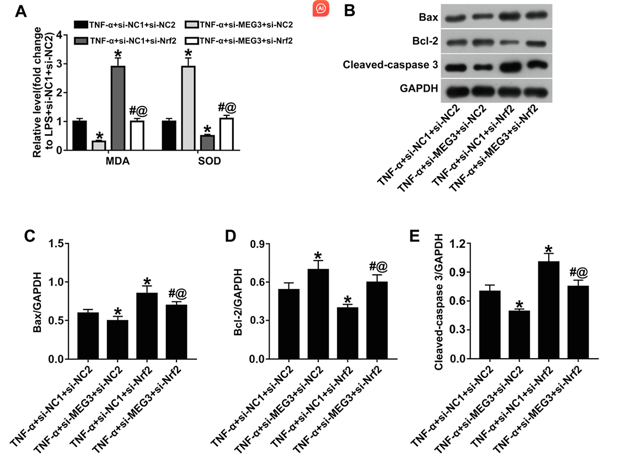
An IC rat model was established by intraperitoneal injection of cyclophosphamide combined with bladder perfusion of fisetin and tumor necrosis factor α using TNF α (TNF α) to mimic IC. Bladder epithelial cells were isolated and cultured in vitro, forming pavement-like sheets after 2-3 weeks. Immunofluorescence confirmed epithelial identity via PCK2 staining (Fig. 1A). TNF-α treatment (50 μg/L, 24 h) mimicked interstitial cystitis conditions, upregulating MEG3 and downregulating Nrf2 expression (Fig. 1B, C). Lentiviral transduction with si-MEG3 upregulated Nrf2, while si-Nrf2 reduced Nrf2 levels (Fig. 1D). Combined si-MEG3+si-Nrf2 treatment reversed the anti-inflammatory effects of si-MEG3 alone, elevating IL-1β, IL-6, and TNF-α (Fig. 1E), increasing oxidative stress markers (ROS, MDA) and reducing SOD (Fig. 1F, G, 2A). Apoptosis was enhanced, with upregulated Bax and cleaved caspase-3 and downregulated Bcl-2 (Fig. 2B-E), confirming Nrf2 mediates MEG3's protective effects against TNF-α-induced damage.
Ask a Question
Write your own review