Human Dermal Fibroblasts from Systemic Lupus Erythematosus (SLE) Patient
Cat.No.: CSC-C9404W
Species: Human
Source: Dermis; Skin
Cell Type: Fibroblast
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Human Dermal Fibroblasts from Systemic Lupus Erythematosus (SLE) Patient cells are primary fibroblast cells cultured from skin tissue obtained from a patient with systemic lupus erythematosus. These cells maintain phenotypic and molecular features associated with disease pathology and serve as an in vitro disease model system for studying the skin and systemic effects of SLE. Under standard cell culture conditions, SLE patient-derived dermal fibroblasts have a normal fibroblast-like morphology and form adherent cell monolayers. They are typically spindle-shaped cells that often proliferate abnormally and show hypersensitivity to inflammatory signals and irregular production of extracellular matrix components when compared to healthy donor-derived fibroblasts. SLE dermal fibroblasts show molecular evidence of upregulated immune- and inflammation-related signaling, such as increased sensitivity to type I interferon and overexpression of interferon stimulated genes.
As such, these cells are used to study fibroblast-immune cell crosstalk, cutaneous inflammation and fibrosis in the context of autoimmune disease. These cells can also be used to model chronic inflammation, tissue remodeling and defective wound healing that occurs in SLE. Additionally, SLE dermal fibroblasts can be used to screen potential anti-inflammatory and immunomodulatory therapeutics.
SLE Dermal Fibroblasts Exhibit Hyperinflammatory Responses
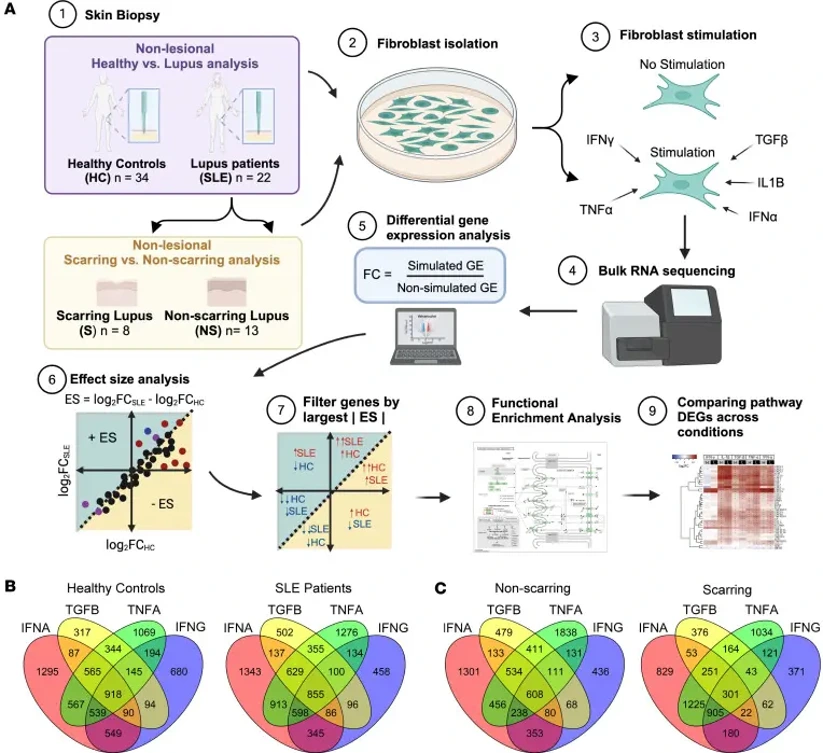
Cutaneous lupus erythematosus (CLE) lesions endure chronic inflammation; however, the mechanisms by which inflammation leads to either scarring or non-scarring disease pathology remain unclear. To gain insight into fibroblast-intrinsic mechanisms that may contribute to tissue pathology, Shoffner-Beck et al. compared cytokine responses between non-lesional dermal fibroblasts isolated from patients with SLE/CLE versus healthy controls.
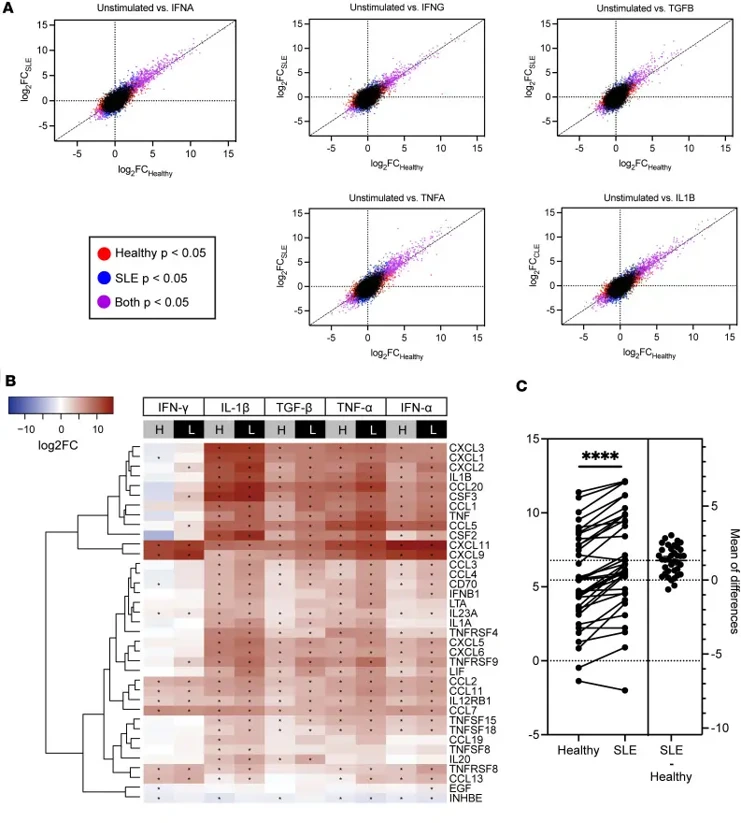
To examine fibroblast functional differences, they established cultures from punch biopsies of healthy control skin and nonlesional lupus skin (upper thigh, non-sun-exposed). Fibroblasts at passage 2 were treated with IFN-γ, IFN-α, TNF-α, TGF-β, or IL-1β (cytokines identified as upstream regulators of differences observed between healthy control and SLE fibroblasts from scRNA-Seq) for 6 h prior to RNA isolation (Fig. 1). Both groups were confirmed to have similar fibroblast populations prior to and after cytokine stimulation. Both healthy control and SLE fibroblasts had differentially expressed genes after cytokine stimulation. However, greatest differences in effect size between healthy and SLE fibroblasts were noted after treatment with TGF-β, TNF-α, IFN-γ, and IFN-α (Fig. 2A). Within cytokine-cytokine receptor signaling pathways, genes belonging to the CXCL family, CCL family, IL family, and TNF had significantly higher expression across all treatments with significantly greater magnitude in SLE fibroblasts (FDR < 1 × 10-5; Fig. 2B). When focusing specifically on responses to TGF-β stimulation, there were significantly higher fold changes between SLE compared to healthy control fibroblasts (Fig. 2C). These data suggest that inflammatory cytokines induce signaling pathways in both healthy and SLE fibroblasts but that this response is exaggerated in SLE fibroblasts.
Ask a Question
Write your own review