Mouse Retinal Müller Cells
Cat.No.: CSC-C5414S
Species: Mouse
Source: Retina; Eye
Cell Type: Glial Cell
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
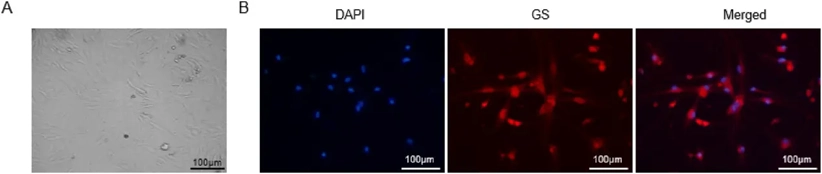
Mouse retinal müller cells from Creative Bioarray are isolated from the mouse eye tissue. The method we use to isolate mouse retinal müller cells was developed based on a combination of established and our proprietary methods. The mouse retinal müller cells are characterized by immunofluorescence with antibodies specific to glutamine synthetase (GS). Each vial contains 0.5x10^6 cells per ml and is delivered frozen.
Mouse Retinal Müller Cells are the principal macroglial elements of the vertebrate retina, spanning the entire thickness of the neural retina from the internal to the external limiting membranes. As the primary support cells, Müller glia are indispensable for maintaining retinal homeostasis, providing structural integrity, and regulating the metabolic and electrophysiological environment of retinal neurons. Our purified primary Mouse Retinal Müller Cells offer a high-fidelity model for investigating retinal physiology, gliosis, and neuroregenerative strategies.
- Essential Homeostatic Mediators: These cells are responsible for critical functions, including potassium spatial buffering (via Kir4.1 channels), neurotransmitter recycling (notably glutamate uptake through GLAST), and the regulation of the blood-retinal barrier. This makes them a superior model for studying diabetic retinopathy and glaucoma.
- Neurogenic Potential & Plasticity: Unlike their human counterparts in most clinical settings, murine Müller cells exhibit a latent "progenitor-like" plasticity. They serve as a vital tool for research into endogenous retinal repair, cellular reprogramming, and the activation of Notch and Wnt signaling pathways for photoreceptor regeneration.
- Robust Secretome Profile: Mouse Müller cells are a prolific source of neurotrophic factors, such as BDNF, CNTF, and VEGF. This secretome is essential for evaluating neuroprotective therapies and understanding the shift from physiological support to pathological "reactive gliosis".
- Preclinical Relevance: Utilizing mouse-derived Müller cells allows for seamless integration with transgenic, knockout, and reporter mouse models (e.g., GFAP-GFP), enabling precise lineage tracing and genetic functional analysis that is unachievable in higher species.
Validated for high purity through the expression of specific markers such as Glutamine Synthetase (GS), Vimentin, and CRALBP, our Mouse Retinal Müller Cells provide a standardized, "assay-ready" platform. They are an indispensable asset for pharmaceutical R&D and academic laboratories focusing on ophthalmology, neurogliology, and the development of next-generation therapies for retinal degenerative diseases.
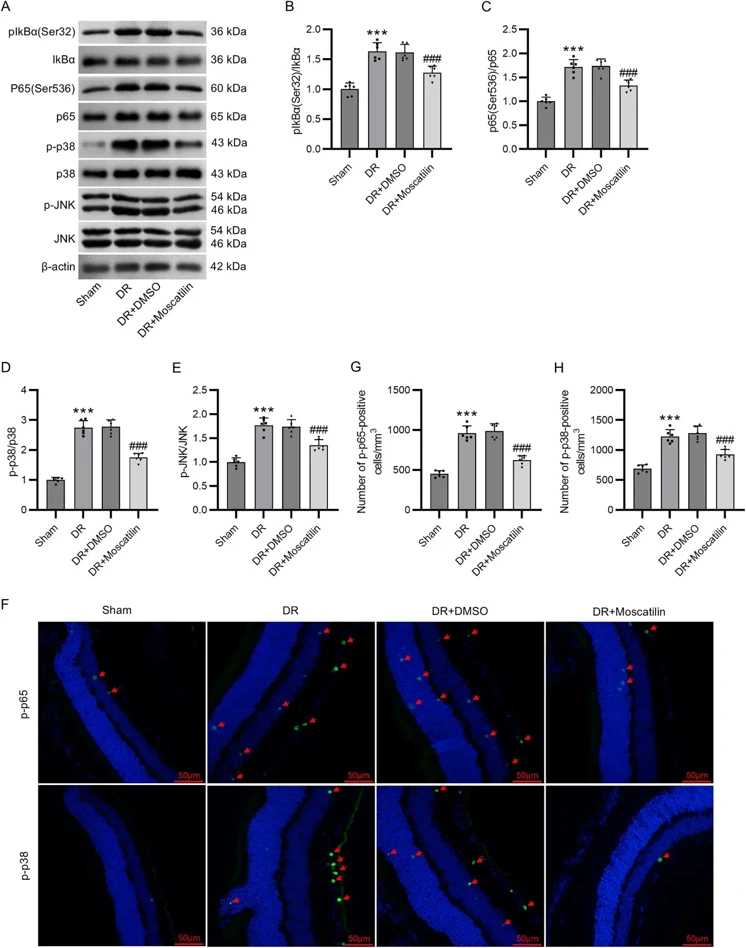
Moscatilin Alleviates Oxidative Stress and Inflammatory Response of Müller Cells in Diabetic Retinopathy Through Suppressing The p38 MAPK/JNK and NF-κB Signaling Pathways
Diabetic retinopathy (DR), as the main ophthalmic complication of diabetes mellitus, is a major eye disorder contributing to blindness. Oxidative stress and inflammation in retinal Müller cells participate in the pathogenesis of DR. This work aims to study the biological role of moscatilin in the progression of DR and the underlying mechanism.
High glucose (HG)-stimulated mouse primary retinal Müller cells and high-fat diet + streptozotocin (STZ)-induced DR mouse models were constructed as in vitro and in vivo models, respectively. The effects of moscatilin treatment on oxidative stress and inflammation in HG-stimulated Müller cells and DR mice were evaluated by detecting intracellular reactive oxygen species production, malondialdehyde levels, superoxide dismutase and catalase activities, glutathione/oxidized glutathione ratio, as well as proinflammatory cytokine levels.
Moscatilin pretreatment prevented HG-induced decrease in Müller cell viability. Moscatilin mitigated oxidative stress, inflammation, and extracellular matrix remodeling in HG-stimulated Müller cells and DR mice. Mechanically, moscatilin reduced the levels of receptor for advanced glycation end products, phosphorylated I-kappa-B-alpha, p-p65 NF-κB, p-p38 MAPK, and p-JNK in both HG-stimulated Müller cells and DR mice. Moscatilin plays an antioxidant and anti-inflammatory role in DR by inhibiting the p38 MAPK/JNK and NF-κB signaling pathways.
Ask a Question
Write your own review