Mouse Peritoneal Mesothelial Cells
Cat.No.: CSC-C5367S
Species: Mouse
Source: Peritoneal Cavity
Cell Type: Mesothelial Cell
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Mouse peritoneal mesothelial cells from Creative Bioarray are isolated from the mouse peritoneal tissue. The method we use to isolate mouse peritoneal mesothelial cells was developed based on a combination of established and our proprietary methods. The mouse peritoneal mesothelial cells are characterized by immunofluorescence with antibodies specific to vimentin or keratin. Each vial contains 0.5x10^6 cells per ml and is delivered frozen.
Derived from the serosal lining of the murine peritoneal cavity, Mouse Peritoneal Mesothelial Cells (MPMCs) are polarized epithelial-like cells that maintain critical barrier, secretory, and immune-surveillance functions. Their primary nature preserves native phenotypic traits lost in transformed lines, offering distinct experimental advantages.
- Pathophysiological Fidelity: MPMCs closely mimic human peritoneal mesothelium, rendering them indispensable for modeling peritoneal dialysis (e.g., ultrafiltration failure, encapsulating sclerosis), post-surgical adhesions, and sterile inflammation.
- Tumor Microenvironment Modeling: They actively interact with disseminated ovarian and gastric cancer cells, providing a unique platform to study transcoelomic metastasis and mesothelial‑to‑mesenchymal transition (MMT).
- Genetic Tractability: When harvested from transgenic or knockout mouse strains, MPMCs enable direct interrogation of gene function in fibrosis, transport, or innate immunity without additional engineering.
- Experimental Versatility: Readily isolated and expanded ex vivo, they retain responsiveness to TGF-β, LPS, and high glucose, supporting mechanistic dissection of signaling pathways and high‑content drug screening.
These attributes establish MPMCs as a superior primary cell system bridging basic peritoneal biology and translational therapeutic development.
Platelet-Rich Fibrin Promotes Mesothelial Cell Proliferation and Migration by Up-Regulating Calretinin to Prevent Postoperative Intestinal Adhesion
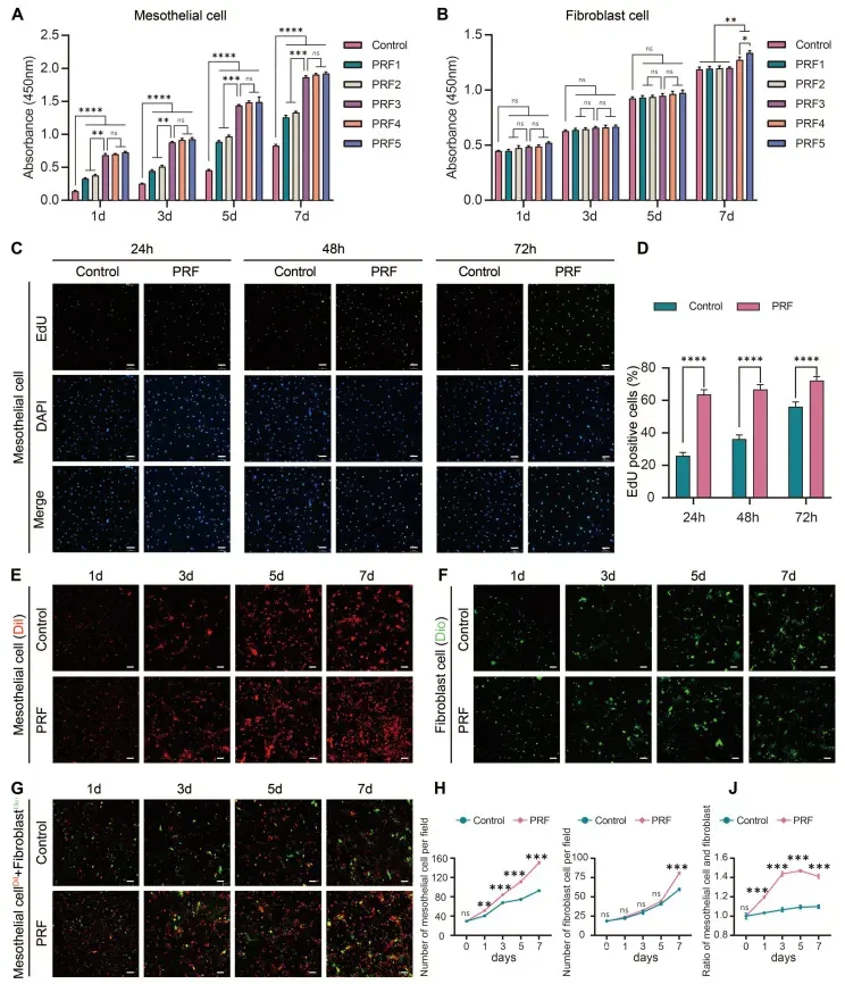
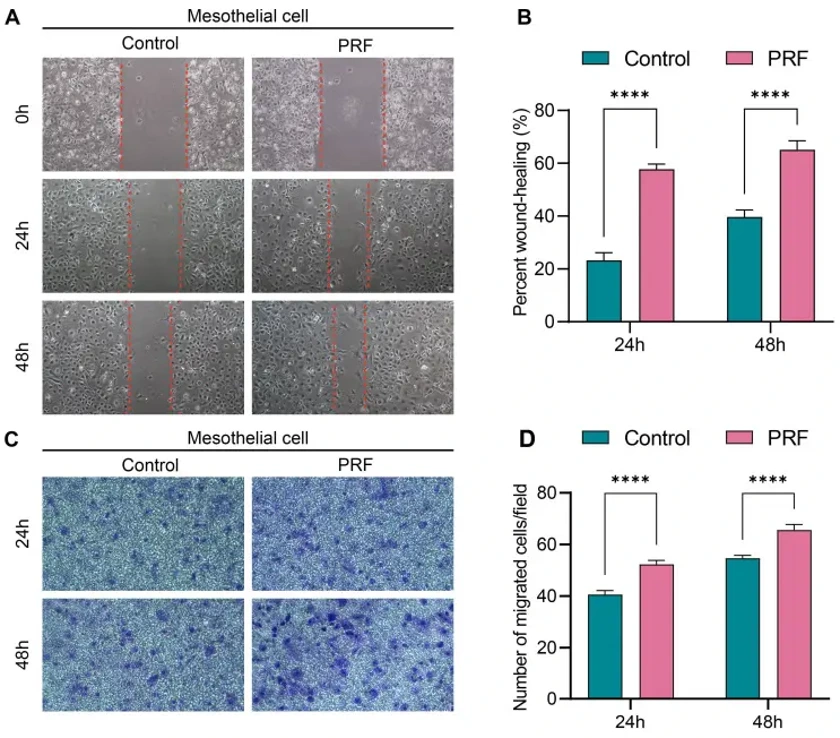
Intestinal adhesions are the most common postoperative complication, leading to intestinal obstruction, female infertility, and even death. The currently developed materials for preventing intestinal adhesions mainly focus on physical barriers and reducing inflammation, while neglecting the importance of effectively promoting rapid repair of the peritoneum. Previous studies have found that platelet-rich fibrin (PRF) can prevent postoperative intestinal adhesions. The proliferation of mesothelial cells may play a significant role in reducing intestinal adhesions, but the mechanism remains unclear.
Primary mouse peritoneal mesothelial cells and mouse peritoneal fibroblasts were used in this study to investigate the effects of PRF on mesothelial cell proliferation, attachment, migration, and mesothelial-mesenchymal transition (MMT). PRF promoted mesothelial cell proliferation from 1st day and significantly promotes fibroblast proliferation from 7th day. Meanwhile, PRF tended to promote the proliferation and attachment of mesothelial cells rather than fibroblasts. However, PRF had a limited regulatory effect on the MMT of mesothelial cells.
Ask a Question
Write your own review