- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Mouse hepatic oval cells from Creative Bioarray are isolated from the mouse liver tissue. The method we use to isolate mouse hepatic oval cells was developed based on a combination of established and our proprietary methods. The mouse hepatic oval cells are characterized by immunofluorescence with antibodies specific to receptor tyrosine kinase (c-Kit) or alpha fetoprotein (AFP). Each vial contains 0.5x10^6 cells per ml and is delivered frozen.
Mouse Hepatic Oval Cells (MHOCs) are bipotential liver progenitor cells isolated from the liver of mice, typically after chronic liver injury or 2/3 partial hepatectomy. These cells serve as an important tool for the study of liver regeneration, hepatic differentiation, and mechanisms of liver disease in vitro because they have the capacity to differentiate into hepatocytes and biliary epithelial cells (cholangiocytes).
MHOCs are small epithelial-like cells that generally proliferate as an adherent monolayer with the ability to cluster into colony-forming masses. They express progenitor cell markers such as A6, EpCAM, CK19 and differentiate to express hepatic markers including alpha-fetoprotein (AFP) and albumin. MHOCs retain responsiveness to growth factors and cytokines such as HGF, EGF and TGF-β important for regulating proliferation, differentiation and survival.
Due to their bipotential differentiation capacity MHOCs are commonly used to study liver regeneration, fibrosis/hepatocarcinogenesis, and cholangiopathies. They have been used as a tool to study mechanisms regulating activation of stem/progenitor cells after chronic liver injury including factors regulating proliferation and migration, signaling pathways important for specification into different cell lineages, drug toxicity testing, and many applications involving gene editing and disease modeling in vitro.
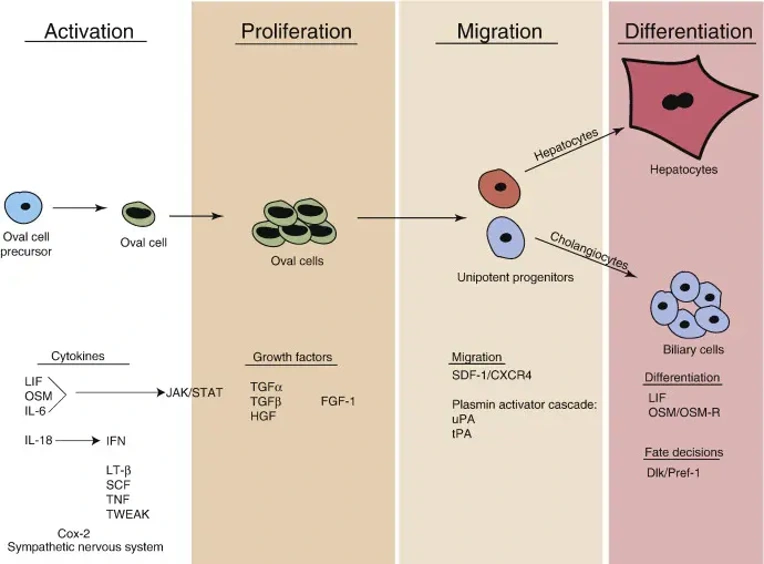
HOCs Morphological Features in the Original Generation and Differentiation into BDEs
Hepatic oval cells (HOCs) can differentiate into cholangiocytes during liver regeneration, but the epigenetic mechanisms governing this transition remain unclear. Jin's team investigated whether histone methyltransferase SETD2 and its product H3K36me3 regulate HOC-to-cholangiocyte differentiation following partial hepatectomy and 2-acetamidofluorene exposure in mice.
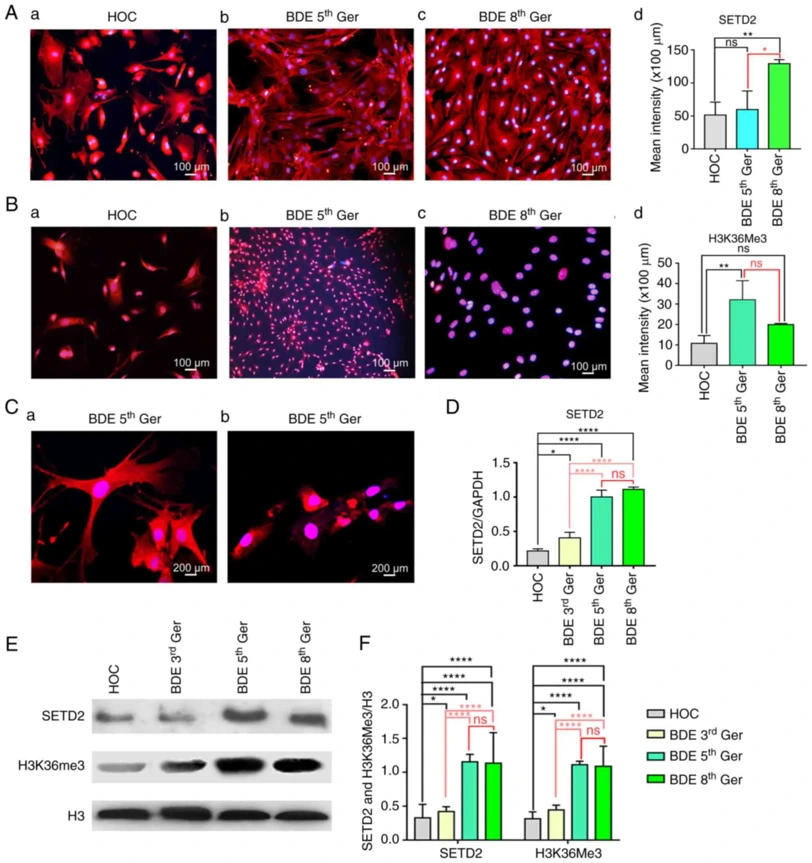
A modified two-step enzyme digestion method established hepatic oval cell (HOC) proliferation in a mouse 2-AAF/PH model. Isolated HOCs were small, oval cells with high nuclear/cytoplasmic ratios (Fig. 1A). After 3 days in cytokine-containing medium, first-generation colonies showed rapid growth with gradually enlarging nuclei (Fig. 1B, C). By passages 2-3, cells became irregular-shaped with large volume and prominent nuclei (Fig. 1D, E). Passages 4-5 showed bile duct epithelial (BDE) differentiation with enlarged cell mass and volume (Fig. 1F, G). Passages 6-8 exhibited bile duct epithelium changes with pseudopodia-like growth and three-dimensional structure without bottom layering (Fig. 1H-J).
They investigated SETD2/H3K36me3 expression changes during HOC differentiation into BDEs. Although SETD2/H3K36me3 roles in cell differentiation are established in other stem cells, their dynamics in this process were previously unexplored. HOCs were induced to differentiate into BDEs using EGF (20 µg/l) + SCF (10 µg/l) + LIF (10 µg/l). Passages 5 and 8, distinguishable by morphology, were selected for analysis. Immunofluorescence showed SETD2 expressed in both nucleus and cytoplasm with significant increase from primary generation onward, while H3K36me3 was mainly nuclear and gradually increased during differentiation (Fig. 2Aa-d, Ba-d). Confocal microscopy of passage 3 BDEs confirmed SETD2 in nucleus/cytoplasm and H3K36me3 predominantly nuclear (Fig. 2Ca, b). RT-qPCR revealed SETD2 mRNA gradually increased and stabilized during differentiation (Fig. 2D). Western blotting showed SETD2 and H3K36me3 protein levels increased ~35% in passage 3 BDEs versus primary HOCs, and ~75% in passages 5 and 8 mature BDEs, thereafter stabilizing (Fig. 2E, F).

Ask a Question
Write your own review