Mouse Lung Microvascular Endothelial Cells
Cat.No.: CSC-C1933
Species: Mouse
Source: Lung
Cell Type: Endothelial Cell; Microvascular Cell
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Mouse Lung Microvascular Endothelial Cells (MLMVECs) are primary endothelial cells obtained from the microvasculature of mouse lung. Pulmonary capillaries are lined by endothelial cells which form an important component of the alveolar-capillary barrier. These cells control gas exchange, vascular permeability and inflammatory response.
MLMVECs display the classic cobblestone morphology of endothelial cells when cultured to confluency. They also express well-established endothelial markers such as CD31 (PECAM-1), VE-cadherin (CD144), von Willebrand factor (vWF) and endothelial nitric oxide synthase (eNOS). Functionally, MLMVECs display contact inhibition of movement, uptake of acetylated LDL and ability to form capillary tube-like structures during angiogenesis assays. These properties establish MLMVECs endothelial phenotype and identify them as microvascular endothelial cells.
MLMVECs are commonly used to study acute lung injury (ALI) and acute respiratory distress syndrome (ARDS), pulmonary hypertension, sepsis-induced endothelial dysfunction, and inflammatory lung disease. They provide an excellent tool to study endothelial barrier regulation, leukocyte adhesion and transmigration, oxidative stress, cytokine networks and angiogenesis. Signaling pathways studied include VEGF signaling, NF-κB, MAPK, PI3K/AKT, and TGF-β signaling.
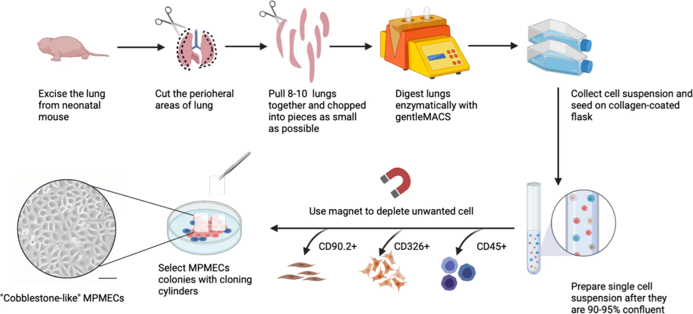
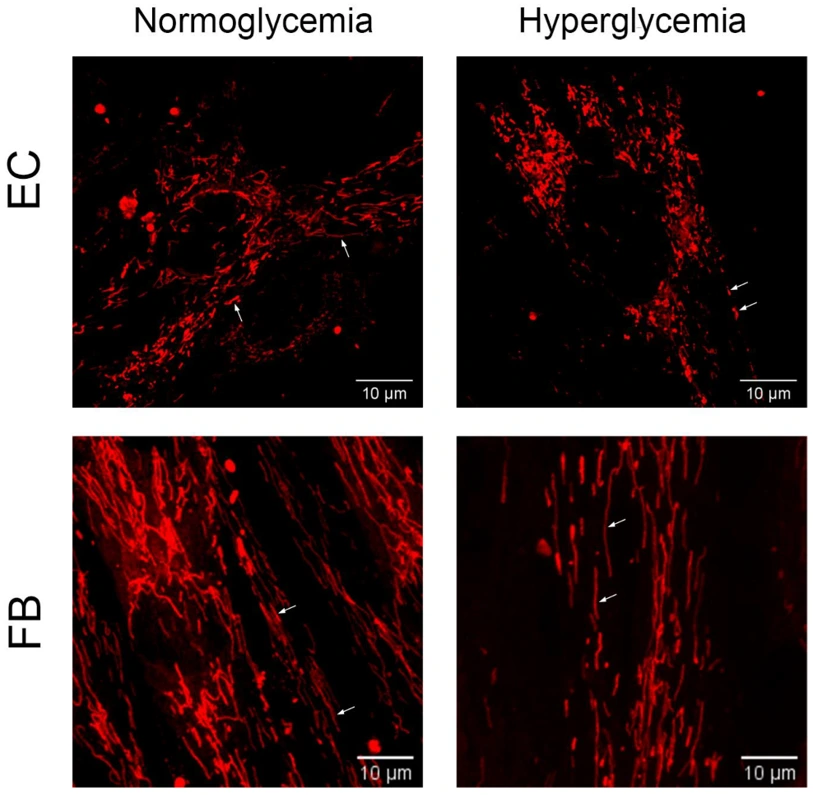
Hyperglycemia Promotes Bidirectional Changes in the Morphology of Individual Mitochondria in Primary Mouse Lung Microvascular Endothelial Cells and Human Dermal Fibroblasts
Belosludtseva et al. investigated the effect of hyperglycemia on mitochondrial morphology in primary mouse lung microvascular endotheliocytes and human dermal fibroblasts. They first found that hyperglycemia induces a decrease in the membrane potential of mitochondria in primary endotheliocytes and fibroblasts. Alterations in mitochondrial mass and morphology are associated with the progression of diabetes and hyperglycemic damage to cells. Therefore, they next analyzed the effect of hyperglycemia on the number and morphological features of the organelles in primary endothelial cells and fibroblasts.
A minimum of 20 images per cell type were captured and evaluated for mitochondrial number, average perimeter, network interconnectivity, and individual elongation. Hyperglycemia caused endotheliocytes mitochondria number to increase by 1.75-fold, perimeter to decrease by 1.12-fold and network interconnectivity decrease by 1.13-fold. These data indicate hyperglycemia caused fragmentation of the endotheliocyte mitochondrial network (Fig. 1). Fibroblasts experienced the opposite effect with mitochondrial number decreasing by 1.3-fold and perimeter increasing by 1.14-fold. Increased individual mitochondrial elongation was also observed; however, network interconnectivity remained consistent. This data shows different cell types respond differently to hyperglycemic environments.
Ask a Question
Write your own review