CD1 Mouse Aortic Endothelial Cells
Cat.No.: CSC-C4232X
Species: Mouse
Source: Aorta
Cell Type: Endothelial Cell
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
CD1 Mouse Aortic Endothelial Cells are primary endothelial cells derived from the aorta of CD1 mice. They offer an in vivo-like representation for investigating vascular biology and endothelial cell function within a controlled laboratory setting. The endothelial cells that line the interior surface of large blood vessels such as the aorta help to maintain vascular permeability, vasomotor tone, inflammation, and hemostasis.
CD1 mouse aortic endothelial cells can be grown to confluency in culture where they demonstrate the cobblestone morphology that is characteristic of endothelial cells. They grow as contact-inhibited monolayers and express classic endothelial markers including CD31 (PECAM-1), VE-cadherin, von Willebrand factor (vWF), and endothelial nitric oxide synthase (eNOS). They also react responsively to a variety of physiological cues as well as pathological stimuli such as shear stress, cytokines, and oxidants. As such, they serve as useful tools for mechanistic studies into endothelial signaling and remodeling. These cells have been used to model human disease such as atherosclerosis and vascular inflammation. They can be used for studies into cardiovascular research, endothelial cell biology, and preclinical therapeutic assessments.
BPA Induced Vascular Injury is Mediated by RIP3/CamKII Dependent Endothelial Cell Necroptosis
Long-term exposure to the xenoestrogen Bisphenol-A (BPA) is epidemiologically linked to adverse cardiovascular outcomes. Reventun et al. previously showed that BPA elevates blood pressure via CaMKII activation and eNOS-uncoupled oxidative stress. Since sustained CaMKII activation can trigger necroptosis, Reventun et al. tested whether BPA-induced coronary endothelial necroptosis drives cardiac dysfunction.
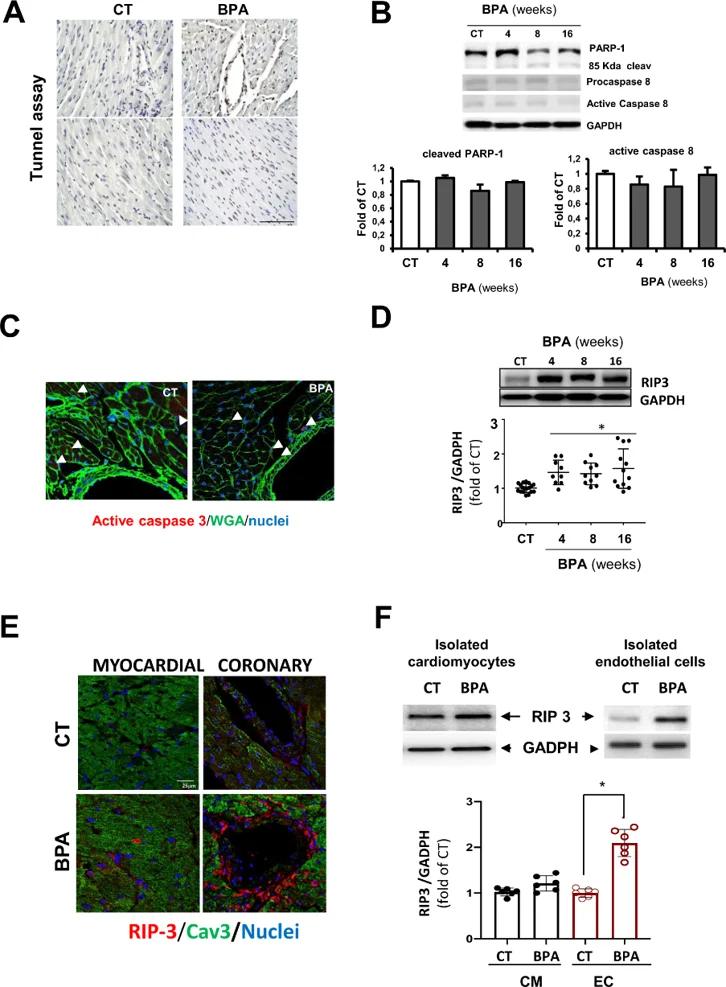
To investigate mechanisms underlying BPA-induced vascular leakage and myocardial hemorrhages, TUNEL assays were performed on hearts from 8-week BPA-treated CD1 mice. Total TUNEL-positive cells increased 2.8-fold, with a 9.9-fold increase in coronary vessel areas, indicating selective vascular susceptibility (Fig. 1A). However, western blot showed no changes in caspase-3 or caspase-8 activity (PARP-1 cleavage; Fig. 1B), and confocal microscopy confirmed minimal active caspase-3 staining (Fig. 1C), suggesting apoptosis-independent cell death. This was validated in vitro: BPA reduced mouse aortic endothelial cell (MAEC) viability from 10-7 M (Fig. 2A) without activating caspase-3 or -8 at 10-6 M (Fig. 2B), whereas cardiomyocytes (H9c2) remained viable except at 10-4 M (Fig. 2C), confirming endothelial-specific, non-apoptotic death. Since caspase-8 negatively regulates necroptosis by cleaving RIP1/3, we examined RIP3 expression. RIP3 was upregulated in BPA-treated hearts at 4, 8, and 16 weeks (Fig. 1D), with intense endothelial localization at 8 weeks (Fig. 1E). Cell fractionation revealed specific RIP3 increase in CD31^+ endothelial cells but not cardiomyocytes (Fig. 1F), and H9c2 cells showed no RIP3 changes (Fig. 2D), indicating BPA activates RIP3-mediated necroptosis selectively in vascular cells.
Ask a Question
Write your own review