C57BL/6 Mouse Resident Peritoneal Macrophages (Frozen Cells)
Cat.No.: CSC-C4239X
Species: Mouse
Source: Peritoneal Cavity
Cell Type: Macrophage
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Mouse Resident Peritoneal Macrophages are negative for bacteria, yeast, fungi, and mycoplasma.Cells can be expanded on a multiwell culture platetandard biochemical procedures performed with cell ready for experiments under the cell culture conditions specified by Creative Bioarray. Repeated freezing and thawing of cells is not recommended.
Mouse Resident Peritoneal Macrophages are tested for expression of markers using antibodies, CD11b by flow cytometry.
Standard biochemical procedures performed with cell cultures include RT-PCR, Western blotting, immunoprecipitation, immunofluorescent staining, flow cytometry or generating cell derivatives for desired research applications.
C57BL/6 Mouse Resident Peritoneal Macrophages are primary immune cells harvested from the peritoneal cavity of C57BL/6 mice. They are used as an in vitro model system that is physiologically relevant to study innate immunity, inflammation, and host-pathogen interactions. These tissue-resident macrophages contribute to homeostasis of the peritoneal cavity by clearing debris and apoptotic cells and mounting immune responses during infection or in response to sterile inflammatory triggers.
These cells can be maintained in culture and demonstrate a typical adherent, round-to-spindle shaped macrophage morphology. They express classic macrophage markers including F4/80, CD11b, and MHC class II indicating their activation state and antigen-presenting properties. C57BL/6 Mouse Resident Peritoneal Macrophages can also be stimulated by a variety of immunological conditions including lipopolysaccharide (LPS), cytokines, and chemokines. Due to their broad range of stimulations and ease of use, they are used to study macrophage polarization, phagocytosis, cytokine production, and activation of signaling pathways.
Baseline Gene Expression in BALB/c and C57BL/6 Peritoneal Macrophages Influences but does not Dictate Their Functional Phenotypes
C57BL/6 and BALB/c mice are reported to skew toward M1 and M2 macrophage phenotypes, respectively, upon pathogen challenge. Using RNA-Seq of unstimulated resident peritoneal macrophages, Restrepo et al. asked whether basal transcriptomes pre-dispose these strains to a given activation state, thereby informing the interpretation of infection studies.
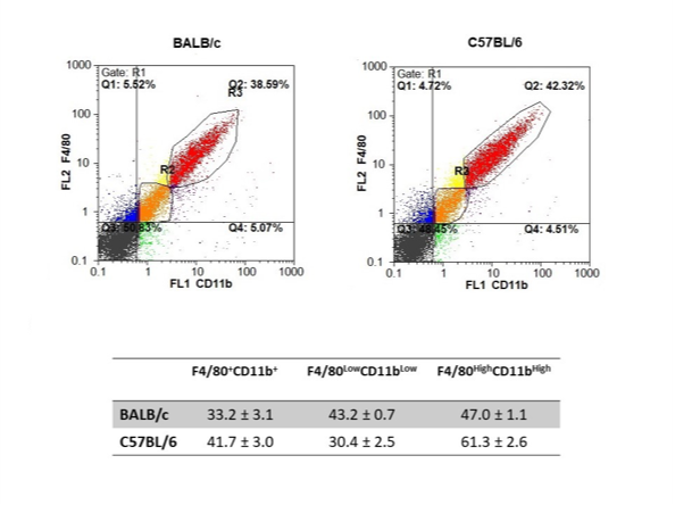
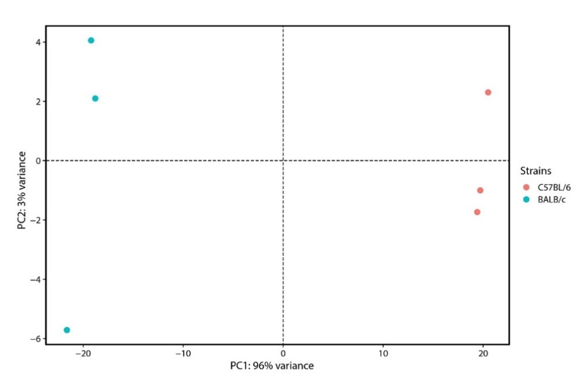
To characterize basal expression differences between BALB/c and C57BL/6 mice, RNA-Seq was performed on unstimulated peritoneal resident macrophages cultured in triplicate. Macrophage purity was confirmed by flow cytometry: CD11b⁺F4/80⁺ cells comprised 33.2 ± 3.1% (BALB/c) and 41.7 ± 3.0% (C57BL/6) of peritoneal lavages, with distinct subset distributions between strains (Fig. 1). Total RNA was extracted after 24 h culture, and poly(A)-enriched sequencing generated ~120 million 150-bp paired-end reads per sample (deposited under BioProject PRJNA656921). On average, 98% of reads mapped unambiguously to the mouse genome. Principal component analysis based on variance-stabilizing transformed data explained 99% of total variance and revealed clear strain-specific clustering (Fig. 2), confirming robust differential expression between BALB/c and C57BL/6 macrophages.
Ask a Question
Write your own review