C57BL/6 Mouse Primary Mammary Fibroblasts
Cat.No.: CSC-C4261X
Species: Mouse
Source: Breast
Cell Type: Fibroblast
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Mouse Primary Mammary Fibroblasts are negative for bacteria, yeast, fungi, and mycoplasma. Cells are tested for expression of marker using the antibody of anti-FSP1/S100A4 by immunofluorescence staining. Cells can be expanded for 3-5 passages at a split ratio of 1:2 under the cell culture conditions specified by Creative Bioarray. Repeated freezing and thawing of cells is not recommended.Standard biochemical procedures performed with cell cultures include the assay of cell to cell interaction, RT-PCR, Western blotting, immunoprecipitation, immunofluorescent staining, flow cytometry or generating cell derivatives for desired research applications.
C57BL/6 Mouse Primary Mammary Fibroblasts are primary stromal cells derived from mammary gland tissue of C57BL/6 mice. They offer an immunologically relevant in vitro model system for studying mammary gland morphogenesis and tumor-stroma interactions. Fibroblasts are a critical component of the mammary gland microenvironment and are responsible for ECM remodeling, secretion of growth factors, and regulation of epithelial cell proliferation and differentiation.
Mouse primary mammary fibroblasts maintain a spindle-shaped fibroblast-like morphology and adhere well to conventional tissue culture surfaces. These cells express typical fibroblast markers such as vimentin, fibronectin, and collagen I, and are negative for epithelial and endothelial markers. Furthermore, mouse primary mammary fibroblasts can become activated to adopt a myofibroblast phenotype when exposed to cytokines/growth factors. Activated myofibroblasts express high levels of α-smooth muscle actin (α-SMA) and have increased ECM production. Derived from the widely used C57BL/6 inbred strain, these primary mammary fibroblasts offer high genetic consistency and compatibility with syngeneic mouse models. They are extensively used to study mammary gland morphogenesis, breast cancer, fibrosis, immune modulation, and epithelial-stromal crosstalk using co-culture.
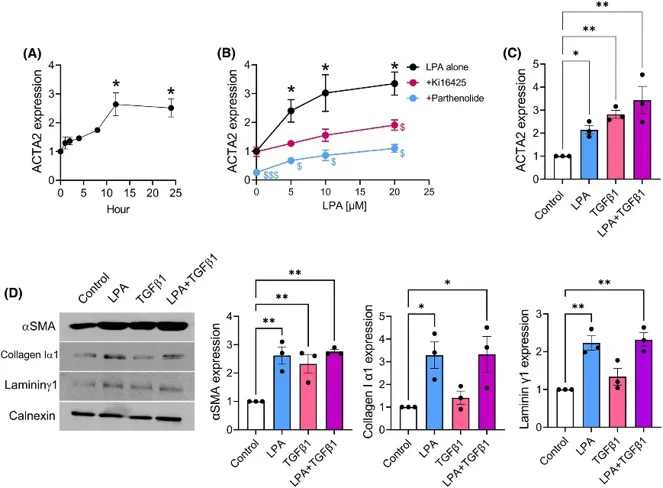
LPA Stimulated Expressions of αSMA, collagen Iα1, and lamininγ1 in Mouse Primary Mammary Fibroblasts
Tumor-associated fibrosis also promotes immunosuppressive conditions that dampen antitumor immunity. Tang et al. sought to determine if IOA-289, an autotaxin (ATX) inhibitor and pioneer in its class, that inhibits production of lysophosphatidate (LPA), had antifibrotic and immune-enhancing properties in breast cancer.
Primary mouse mammary fibroblasts were isolated from the mammary glands of C57BL/6J mice and validated for fibroblast identity by significantly higher expression of FAP, αSMA (ACTA2), and S100A4 when compared to E0771 breast cancer cells. LPA stimulated ACTA2 mRNA expression in a time- and dose-dependent manner. Specifically, 5 μM LPA induced ~2.5-fold and ~2.6-fold upregulation of ACTA2 at 12 h and 24 h, respectively (Fig. 1A). Pre-treatment with the LPAR1/3 inhibitor Ki16425 (0.5 μM) or NF-κB inhibitor parthenolide (5 μM) inhibited LPA-induced ACTA2 expression (Fig. 1B). Treatment with TGFβ1 (10 ng/mL, 24 h) also stimulated ACTA2 expression, but no significant interaction was detected when combined with LPA treatment (Fig. 1C). Both LPA and TGFβ1 upregulated αSMA protein levels at 48 h (Fig. 1D). Of note, LPA significantly upregulated expression of collagen Iα1 and lamininγ1, major ECM proteins, while treatment with TGFβ1 had no effect on either protein expression.
Ask a Question
Write your own review