C57BL/6 Mouse Brain Microvascular Endothelial Cells
Cat.No.: CSC-C1862
Species: Mouse
Source: Brain
Cell Type: Endothelial Cell; Microvascular Cell
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
C57BL/6 Mouse Brain Microvascular Endothelial Cells (mBMVECs) represent the most sophisticated in vitro tool for recapitulating the physiological complexities of the Blood-Brain Barrier (BBB). Derived from the highly characterized and genetically stable C57BL/6 strain, these cells are essential for researchers investigating neuroinflammation, stroke, and the transport kinetics of CNS-targeted therapeutics.
- Exceptional Barrier Function: Our mBMVECs exhibit high expression of critical tight junction proteins, including ZO-1, Occludin, and Claudin-5. This results in superior Trans-Endothelial Electrical Resistance (TEER) values, providing a robust and restrictive barrier for permeability assays.
- Strain-Specific Consistency: By utilizing the C57BL/6 lineage-the industry standard for transgenic and knockout models-our cells ensure seamless data integration with in vivo studies, minimizing genetic drift and maximizing experimental reproducibility.
- Functional Transport Systems: These cells maintain polarized expression of key efflux transporters (e.g., P-gp/ABCB1, BCRP) and nutrient transporters (e.g., GLUT-1), making them an ideal platform for studying drug efflux and receptor-mediated transcytosis.
- Purity and Phenotypic Stability: Rigorously validated for endothelial markers like CD31 (PECAM-1) and vWF, our mBMVECs are guaranteed free from contamination by pericytes or astrocytes, ensuring that your results reflect pure microvascular responses.
Elevate your CNS drug discovery pipeline with mBMVECs that offer the structural integrity and metabolic fidelity required for high-impact neurovascular research.
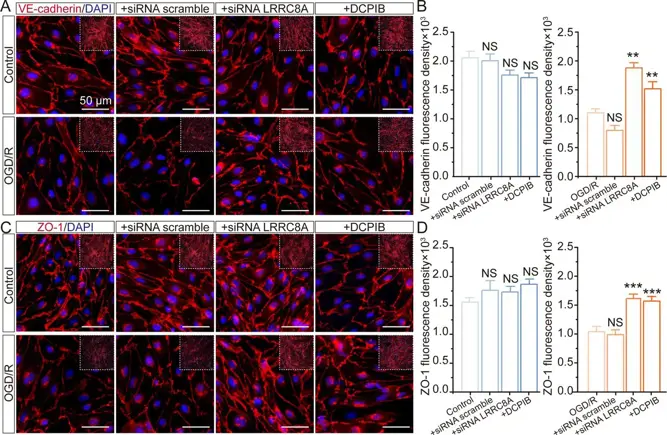
LRRC8A in Endothelial Cells Contributes to The Aberrant Blood-Brain Barrier Integrity in Ischaemic Stroke
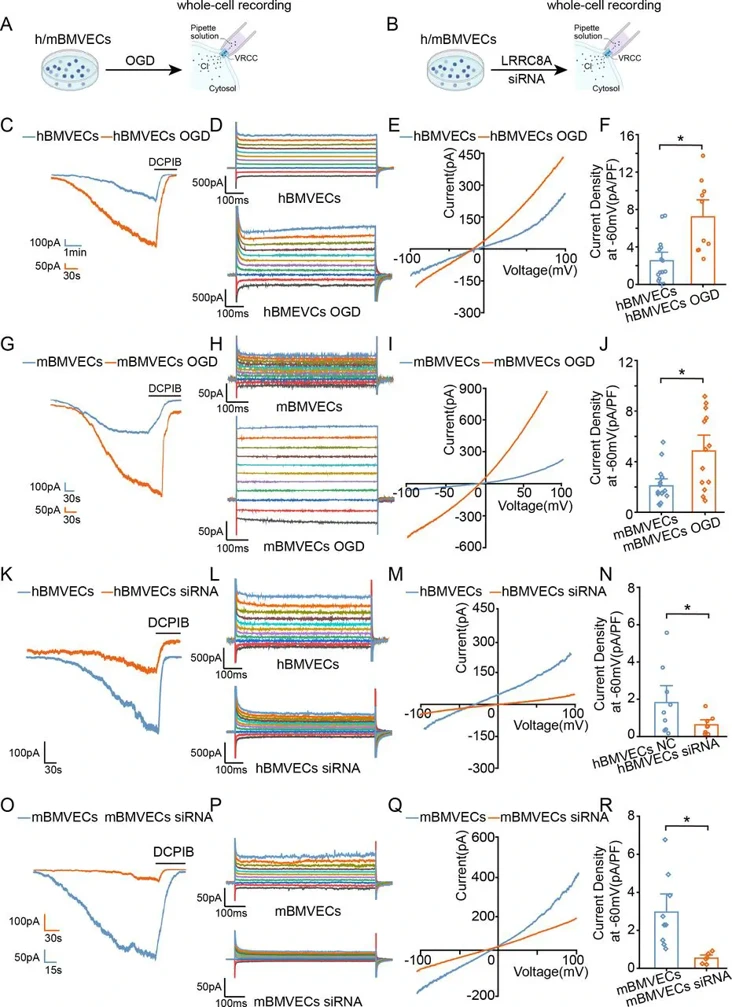
The increased permeability of the blood-brain barrier (BBB) is a critical contributor to the high mortality following ischaemic stroke. However, the mechanisms regulating BBB integrity remain poorly understood. Leucine-rich repeat-containing 8A (LRRC8A) is a chloride channel critical for cellular volume homeostasis and plays a key role in regulating neuronal injury during ischaemia. However, its impact on BBB function is currently unclear.
Primary mouse and human brain microvascular endothelial cells (m/hBMVECs) were subjected to oxygen-glucose deprivation (OGD) and re-oxygenation for varying durations. Patch-clamp recordings were performed to measure volume-regulated chloride currents. Immunostaining was conducted to evaluate protein expression. Cell permeability was evaluated with transwell assay.
OGD exposure augments the Volume-regulated chloride channel (VRCC) current mediated by LRRC8A in BMVECs. In contrast, inhibiting LRRC8A promotes the expression of ZO-1 and VE-cadherin, thereby preserving the integrity of endothelial cells. With-no-lysine kinase 1 (WNK1) inhibition contributes to LRRC8A-induced BBB damage post-ischaemic stroke. Eupatorin, a newly identified LRRC8A inhibitor, exerts neuroprotective effects against ischaemic stroke.
Ask a Question
Write your own review