Immortalized Human Nasopharyngeal Epithelial Cells (NPEC2/Bmi1)
Cat.No.: CSC-I9353L
Species: Homo sapiens
Source: Nasopharynx
Morphology: Cobblestone-like
Culture Properties: Adherent
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Note: Never can cells be kept at -20 °C.
CIK-HT003 HT® Lenti-SV40T Immortalization Kit
Immortalized Human Nasopharyngeal Epithelial Cells (NPEC2/Bmi1) are an immortalized cell line derived from normal human nasopharyngeal epithelial cells that stably express Bmi1, a polycomb group protein that has been shown to prolong cellular lifespan by modulating cell cycle control and senescence pathways. They represent an immortalized epithelial counterpart to the majority of tumor-derived cell lines, retaining many features of non-malignant nasopharyngeal epithelium but with the added benefit of practical, long-term stable growth.
NPEC2/Bmi1 cells maintain key aspects of epithelial identity. Morphologically, the cells display typical cobblestone-like epithelial appearance, and express many markers of nasopharyngeal epithelial differentiation as well as cell-cell junctions. Bmi1-mediated immortalization was shown to avoid replicative senescence without strong oncogenic changes, making this cell line a stable intermediate model between primary cells and tumor-derived cancer cell lines.
Researchers often use this cell line to explore nasopharyngeal epithelial biology and early stages of NPC development alongside host-pathogen dynamics with a focus on Epstein-Barr virus (EBV) infection. In particular, NPEC2/Bmi1 cells provide a non-tumor background and genetic stability that has been used to study epithelial transformation and signaling pathway dysregulation in the nasopharynx, as well as to test preventive or therapeutic strategies for nasopharyngeal diseases in early stages.
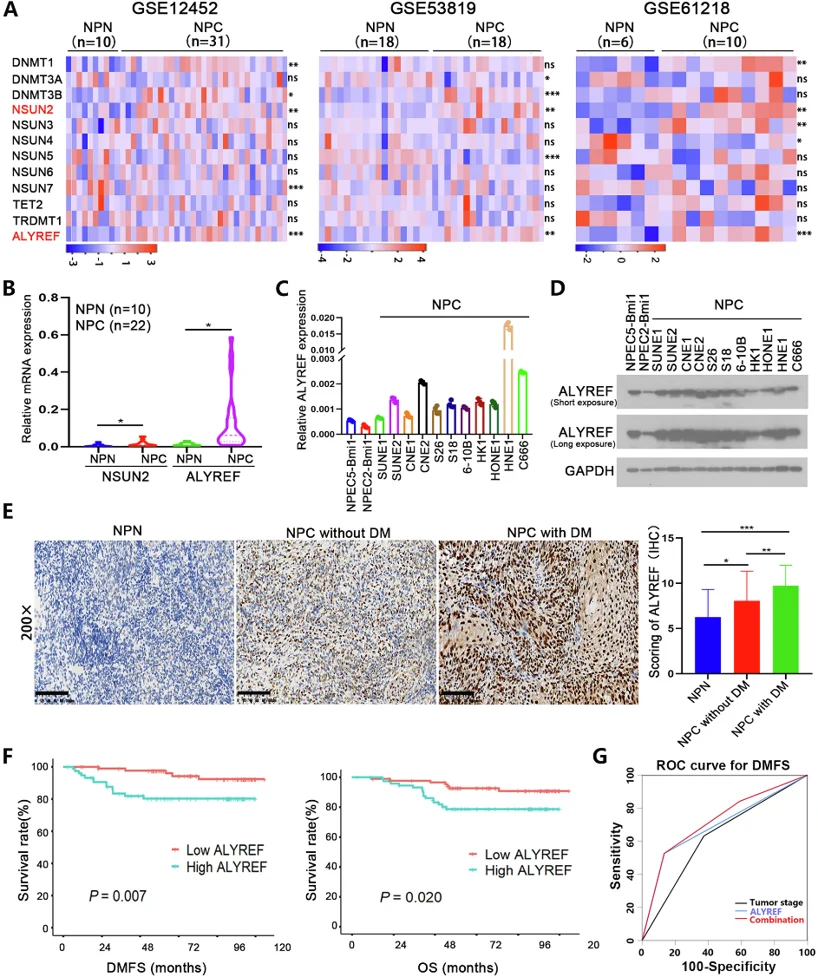
ALYREF is Upregulated in NPC Tissues And Associated with Poor Prognosis in NPC Patients
Approximately 70% of treatment failures in nasopharyngeal carcinoma (NPC) are due to distant metastasis, but the underlying mechanisms are unclear. RNA 5-methylcytosine (m5C) modification controls gene expression and is critical in tumor progression. Jin's team investigated the role of the m5C reader ALYREF in NPC metastasis.
To study the differential expression of m5C-related genes in NPC, Jin's team analyzed three GEO microarray datasets (GSE12452, GSE53819, GSE61218) comparing NPC and non-NPC tissues. NSUN2 and ALYREF were significantly upregulated in NPC tissues (Fig. 1A). Analysis of 22 NPC and 10 non-NPC clinical samples also showed upregulation of NSUN2 and ALYREF in NPC tissues (Fig. 1B), matching the GEO dataset results. NSUN2 has been linked to distant metastasis in NPC, while the role of ALYREF, an RNA m5C reader protein, in NPC is largely unknown. They then examined ALYREF expression in 11 NPC and 2 non-malignant epithelial cell lines (NPEC2-Bmi1 and NPEC5-Bmi1). qRT-PCR (Fig. 1C) and western blot analysis (Fig. 1D) confirmed ALYREF upregulation in NPC cell lines. These findings suggest that ALYREF may contribute to NPC progression.
Ask a Question
Write your own review
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells