Immortalized Human Retinal Pigment Epithelial Cells
Cat.No.: CSC-I9155L
Species: Homo sapiens
Source: Retina
Morphology: Elongated and fusiform shaped
Culture Properties: Adherent
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Note: Never can cells be kept at -20 °C.
CIK-HT013 HT® Lenti-hTERT Immortalization Kit
CIK-HT003 HT® Lenti-SV40T Immortalization Kit
Immortalized Human Retinal Pigment Epithelial Cells (RPE-Im) are human retinal pigment epithelial cells that have been genetically manipulated to bypass replicative senescence but retain characteristics of native RPE. Cultured RPE-Im cell lines have been shown to display a polarized, cobblestone phenotype, grow as tight junction-associated monolayers expressing ZO-1, occludin, and claudins, express lineage specific markers including RPE65, CRALBP (RLBP1), bestrophin-1 (BEST1), MITF, cytokeratins 8/18, phagocytose photoreceptor outer segments, secrete VEGF and PEDF in a polarized manner and participate in the visual cycle, all characteristics of native RPE cells. Several immortalized lines are capable of developing TER and vectorial transport making them useful for blood-retinal barrier studies.
Immortalized human RPE cells have been utilized to study AMD, diabetic retinopathy, oxidative stress, complement activation, mitochondrial dysfunction and inflammation-mediated retinal degeneration as well as serve as a platform for gene therapy vectors, drug permeability across the outer blood-retinal barrier and retinal toxicology assays.
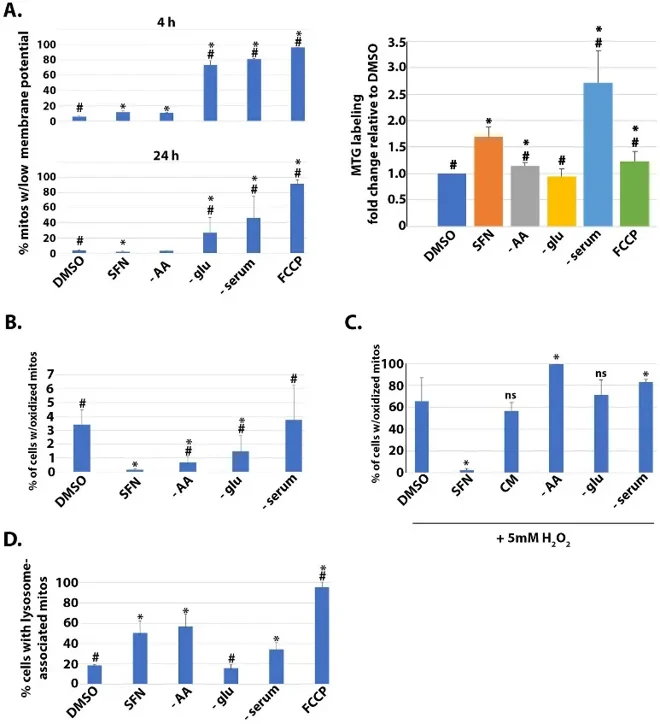
Effects of SFN on Mitochondrial Membrane Potential, Mass, Matrix Redox Status, and Mitophagy
Sulforaphane (SFN) from cruciferous vegetables shows anti-cancer, anti-microbial, and anti-oxidant effects, similar to dietary restriction benefits. Plafker et al. tested whether SFN acts as a fasting/caloric restriction mimetic by examining its effects on nutrient-sensing pathways in human retinal pigment epithelial cells.
They first compared SFN effects versus nutrient deprivations on mitochondrial mass and membrane potential in immortalized human retinal pigment epithelial cells (RPE-1 cells) using MitoTracker Green (MTG) and TMRE labeling with flow cytometry. FCCP served as positive control for membrane potential loss (Fig. 1A). SFN caused modest membrane potential decrease at 4h but preserved potential and increased mitochondrial mass at 24h, closely resembling amino acid (AA) deprivation. Glucose deprivation reduced membrane potential, while serum deprivation increased mass but reduced potential (Fig. 1A). Using mito-roGFP to assess mitochondrial matrix oxidation, SFN suppressed oxidized mitochondria at 4h (Fig. 1B). Both AA and glucose starvation also decreased oxidation, though less than SFN, with AA starvation most similar to SFN. Given SFN's resistance to mitochondrial oxidation, they tested protection against exogenous oxidative challenge. SFN pre-treatment prevented H₂O₂-induced mitochondrial oxidation, unlike nutrient deprivations (Fig. 1C). Using mtKeima-parkin RPE-1 cells to measure mitophagy, SFN, AA starvation, and serum deprivation increased mitophagic flux, while glucose deprivation showed no effect (Fig. 1D). Together, SFN preserves mitochondrial membrane potential, limits matrix oxidation, and promotes homeostatic mitophagy, most closely mirroring AA starvation.
Ask a Question
Write your own review
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells
