Immortalized Human Hepatocytes-Ras
Cat.No.: CSC-I9020L
Species: Homo sapiens
Source: Liver
Morphology: Polygonal
Culture Properties: Adherent
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Note: Never can cells be kept at -20 °C.
CIK-HT025 HT® Lenti-Ras V12 Immortalization Kit
Immortalized Human Hepatocytes (Ras-transformed) represent a cutting-edge in vitro platform designed to overcome the critical limitations of primary human hepatocytes (PHHs), such as rapid loss of metabolic function and limited proliferative capacity. This cell line is established through the stable expression of the oncogenic Ras(e.g., H-Ras), often in combination with other immortalization factors (like hTERT or SV40 T-antigen), to ensure robust, indefinite expansion while maintaining essential hepatic phenotypes.
- Exceptional Proliferative Robustness: Unlike PHHs, which suffer from early senescence, the Ras-transformed line provides an inexhaustible cell source. This scalability ensures lot-to-lot consistency, reducing experimental variability and operational costs for large-scale screening.
- Maintenance of Hepatic Fidelity: These cells retain fundamental hepatocyte characteristics, including the expression of key transcription factors (HNF4α), synthesis of albumin, and crucial phase I/II metabolic enzyme activities. This makes them a superior alternative to hepatocarcinoma-derived lines (like HepG2) which often lack mature metabolic profiles.
- Enhanced Metabolic and Signaling Studies: The integration of the Ras signaling pathway provides a unique model for investigating Ras-MAPK driven hepatocarcinogenesis, intracellular signaling transduction, and liver regeneration mechanisms.
- Optimized for Drug Discovery: The cell line serves as a high-fidelity platform for high-throughput screening (HTS) in drug-induced liver injury (DILI) assays, viral hepatitis research (HBV/HCV), and metabolic disease modeling (NAFLD/NASH).
Our Immortalized Human Hepatocytes-Ras offer a powerful, reproducible, and cost-effective solution for pharmaceutical R&D and academic research. By bridging the gap between unstable primary cultures and undifferentiated tumor lines, this product is an essential tool for accelerating the development of next-generation hepatology therapeutics and diagnostic strategies.
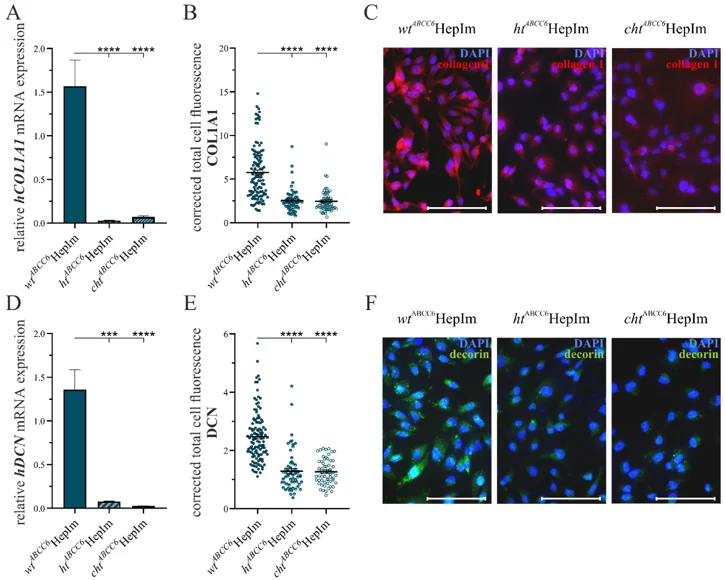
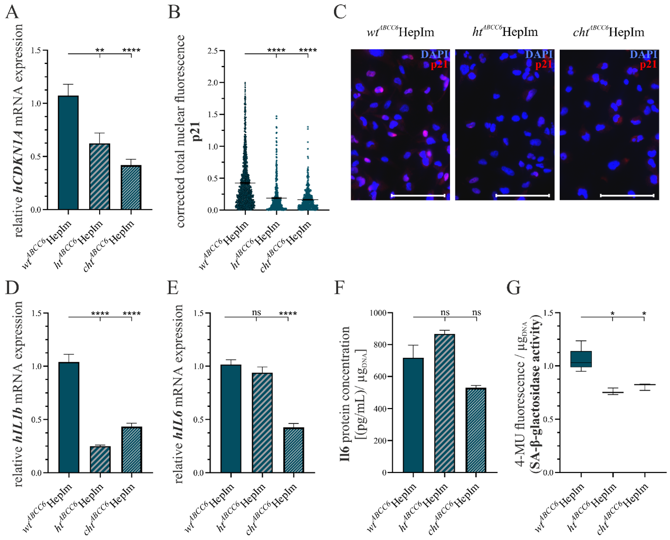
ABCC6-Deficient Human Hepatocytes for Studying the Metabolic Characteristics of PXE
Patients affected by the rare disease pseudoxanthoma elasticum (PXE) exhibit the calcification of elastic fibers in ocular, dermal, and vascular tissues. These symptoms are triggered by mutations in the ATP-binding cassette transporter subfamily C member 6 (ABCC6), whose substrate remains unknown. Interestingly, ABCC6 is predominantly expressed in the liver tissue, leading to the hypothesis that PXE is a metabolic disorder. Plümers, Ricarda developed a genome-editing system targeting ABCC6 in human immortalized hepatocytes (HepIms) for further investigations. The HepIms were transfected with an ABCC6-specific clustered regulatory interspaced short palindromic repeat (CRISPR-Cas9) genome-editing plasmid, resulting in the identification of a heterozygous (htABCC6HepIm) and a compound heterozygous (chtABCC6HepIm) clone. These clones were analyzed for key markers associated with the PXE pathobiochemistry. Hints of impaired lipid trafficking, defects in the extracellular matrix remodeling, the induction of calcification inhibitor expression, and the down regulation of senescence and inflammatory markers in ABCC6-deficienct HepIms were found. The ABCC6 knock-out model of HepIms provides a valuable tool for studying the metabolic characteristics of PXE in vitro. The initial analysis of the clones mirrors various features of the PXE pathobiochemistry and provides an outlook on future research approaches.
Ask a Question
Write your own review
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells