Immortalized Human Hepatocytes-Myc
Cat.No.: CSC-I9019L
Species: Homo sapiens
Source: Liver
Morphology: Polygonal
Culture Properties: Adherent
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Note: Never can cells be kept at -20 °C.
CIK-HT024 HT® Lenti-Myc T58A Immortalization Kit
Immortalized human hepatocytes (IHHs) constitute a rationally engineered alternative cell source positioned strategically between the gold-standard primary human hepatocytes (PHHs) and hepatoma-derived lines. Their scientific advantage is dual: they overcome the critical supply bottleneck of PHHs-plagued by scarcity, donor variability, and rapid functional dedifferentiation-while avoiding the metabolic inadequacy and tumorigenic background endemic to HepG2/Huh7 models.
Functionally superior to carcinoma lines - Unlike HepG2, IHHs retain inducible cytochrome P450 (CYP3A4, CYP2C, CYP1A2) and authentic acute-phase response (IL-6-stimulated fibrinogen upregulation with reciprocal albumin modulation), demonstrating hepatic polarity with bile canaliculi-like vacuoles and correct localization of efflux transporters (MDR1, MRP2). Select lines (HepZ, HepLL) exhibit albumin secretion and ureagenesis at levels statistically indistinguishable from short-term PHH cultures.
Engineered for biorelevance - IHHs demonstrate exceptional tolerance to pathophysiological insults-HepLi-2 cells sustain CYP3A4/2E1 expression and ammonia/lidocaine clearance after 24 h exposure to acute-on-chronic liver failure plasma, a stress lethal to many hepatoma lines. In 3D radial-flow bioreactors, OUMS-29 cells increase albumin output 6-fold over monolayer and sustain CYP3A4-mediated metabolism beyond 29 days, directly enabling chronic toxicology and drug screening paradigms impossible with PHH.
Thus, IHHs represent not merely a PHH surrogate, but a mechanistically tractable, scalable, and increasingly safe platform for pharmacotoxicology, bioartificial liver engineering, and human-relevant disease modeling where PHH are unobtainable and carcinoma cells unreliable.
Panduratin A from Boesenbergia Rotunda Suppresses Hepatitis B Virus by Targeting HNF1α
Boesenbergia rotunda (fingerroot) is widely used in traditional medicine, and its bioactive compound panduratin A has demonstrated potent antiviral properties. However, the mechanistic basis underlying its anti-hepatitis B virus (HBV) activity remains to be fully elucidated.
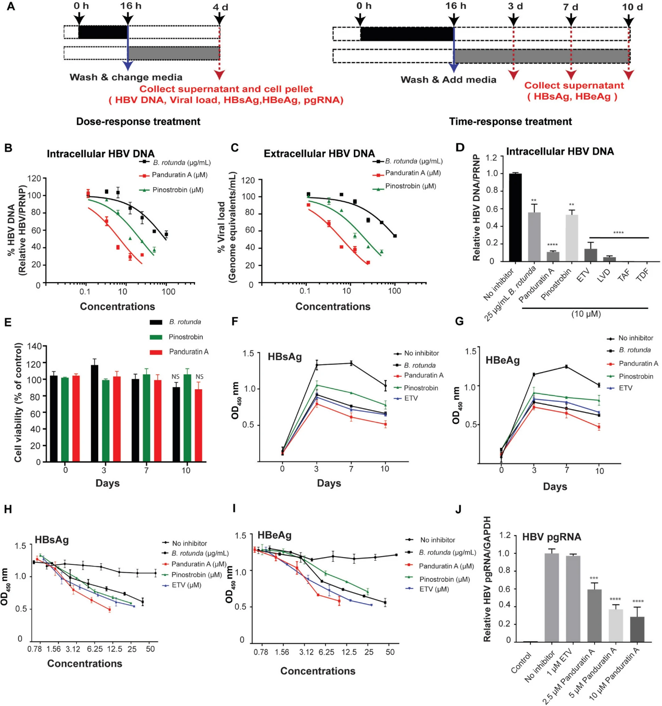
HBV-infected human hepatocytes (imHCs) were treated with B. rotunda extract, panduratin A, or pinostrobin. Intracellular HBV DNA, secreted HBsAg and HBeAg, and pregenomic RNA (pgRNA) were quantified in dose- and time-dependent experiments.
The results showed that crude extract of Boesenbergia rotunda and its purified compounds, panduratin A and pinostrobin inhibited the replication of HBV. These compounds effectively decreased HBV viral load, intracellular HBV DNA, and the levels of HBV proteins, including HBsAg, HBeAg, and HBcAg, comparable to direct-acting antiviral drugs (DAAs). Particularly, panduratin A exhibited superior anti-HBV activity, reducing HBV DNA levels more effectively than entecavir. The inhibitory effects of panduratin A involved in downregulating HNF1α, thereby inhibiting HBV transcription and replication through modulation of the HNF1α axis.
Ask a Question
Write your own review
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells