Immortalized Human Fetal Osteoblast-SV40
Cat.No.: CSC-I9021L
Species: Homo sapiens
Source: Bone
Morphology: polygonal
Culture Properties: Adherent
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Note: Never can cells be kept at -20 °C.
CIK-HT003 HT® Lenti-SV40T Immortalization Kit
Immortalized Human Fetal Osteoblast‑SV40 (hFOB1.19‑SV40) cells are human osteoblastic cells immortalized with the SV40 large T antigen. These cells were originally derived from fetal human bone and provide a conditionally proliferative osteoblastic cell line. The parental hFOB1.19 cell line was immortalized through transfection with the SV40 T antigen which is temperature sensitive to allow for proliferation while maintaining osteoblastic phenotypic characteristics (Keller et al., 1994). Primary osteoblasts isolated from tissues can only survive for a limited amount of time in vitro. Therefore, immortalized cells like hFOB1.19 cells allow for a more robust and reproducible osteoblastic model system.
The hFOB1.19‑SV40 cells are polygonal in shape with a fibroblast-like morphology and grow as adherent monolayers. They express osteoblast specific markers such as alkaline phosphatase (ALP), osteocalcin, bone sialoprotein and type I collagen, further confirming osteoblastic characteristics. When treated with differentiation media containing ascorbic acid and β‑glycerophosphate cells treated showed increased ALP activity and mineralization of the matrix, indicating the cells maintain osteogenic differentiation potential.
Immortalized human fetal osteoblast cells have been used to study osteoblast proliferation and differentiation, regulation of the extracellular matrix, mechanotransduction, evaluation of biomaterials, drug screening assays, bone remodeling as well as response to hormonal regulation such as vitamin D and parathyroid hormone.
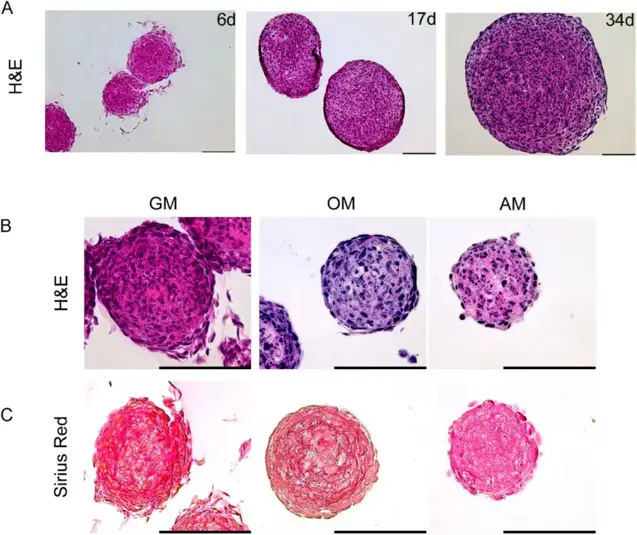
Human Fetal Osteoblast Cells (hFOB) form Solid Spheroids under Proliferative Conditions in the Absence of a Necrotic Core
Skeletal bone function depends on cells and their niches, which together regulate differentiation and remodeling. Here, Marozin et al. present an in vitro 3D model using human fetal osteoblasts (hFOB 1.19) to study osteocyte commitment and examine effects of biomaterials or substances on these processes.
hFOB 1.19 cells form spheroids in non-adherent 3D culture. Histological analysis showed spheroids grown >30 days under proliferative conditions exhibited no necrosis (Fig. 1A), confirmed across osteogenic and adipo-inductive conditions. Proliferative spheroids continued growing, while induction media caused growth arrest and nuclear fragmentation (Fig. 1B). Adipogenesis-induced spheroids were small with irregular boundaries and unstructured appearance, lacking shell-core distinction. Osteogenesis-induced spheroids showed minimal growth and smaller size than growth medium controls, with H&E staining revealing a basophilic environment and no necrotic cores. Growth medium spheroids displayed strong eosinophilic staining compared to induction media. Sirius red staining for collagen visualization (Fig. 1C) showed structured extracellular matrix throughout growth medium spheroids, with highest density in central regions. Osteogenic spheroids exhibited ring-shaped, concentric matrix layers between deep shell cells, while adipogenic spheroids showed no characteristic collagen staining.
Ask a Question
Write your own review
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells
