Immortalized Human Dermal Microvascular Endothelial Cells
Cat.No.: CSC-I9095L
Species: homo sapiens
Source: Skin
Morphology: Polygonal
Culture Properties: Adherent
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Note: Never can cells be kept at -20 °C.
CIK-HT013 HT® Lenti-hTERT Immortalization Kit
CIK-HT003 HT® Lenti-SV40T Immortalization Kit
free from contaminations (bacteria incl. mycoplasma, fungi, HIV, HAV, HBV, HCV, Parvo-B19) and cross-contaminations
Immortalized Human Dermal Microvascular Endothelial Cells (iHDMECs) are immortalized endothelial cells of microvascular origin derived from human tissue. They maintain important endothelial properties and are useful as an in vitro model system to study microvascular biology, angiogenesis and endothelial cell function. One of the most commonly used immortalized human dermal microvascular endothelial cell lines is called HMEC‑1. HMEC‑1 cells were initially isolated by infecting primary human dermal microvascular endothelial cells with an SV40 large T antigen-containing retroviral vector.
Histologically, iHDMECs are classic cobblestone‑like endothelial cells that form adherent monolayers. Like other endothelial cells, iHDMECs express classic endothelial markers such as CD31 (PECAM‑1), VE‑cadherin, von Willebrand factor (vWF), intercellular adhesion molecule‑1 (ICAM‑1) and vascular cell adhesion molecule‑1 (VCAM‑1). These adhesion molecules can be upregulated by treatment with inflammatory stimuli. iHDMECs also retain lipoprotein uptake and cytokine responsiveness. They can be used as an alternative model to study inflammation and lipid-endothelial cell interactions. iHDMECs have been shown to be a useful alternative to primary human dermal microvascular endothelial cells to study endothelial physiology including leukocyte adhesion, barrier function, and angiogenesis.
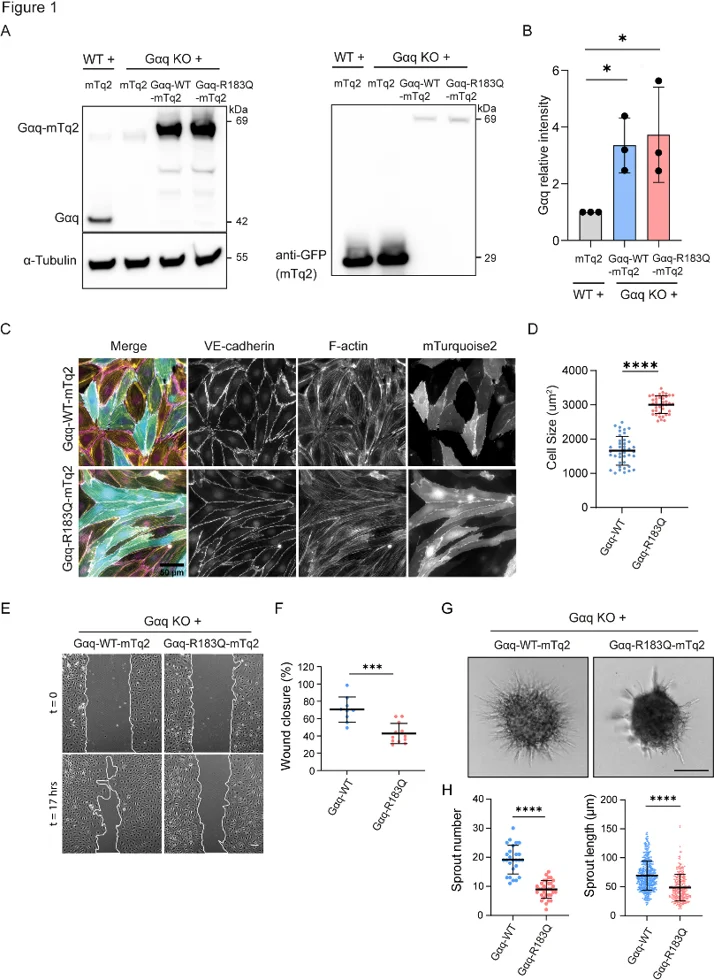
Gαq-R183Q Impairs Endothelial Cell Migration and Angiogenic Sprouting
Capillary malformations (CMs) are caused by somatic mutations in GNAQ (p.R183Q) within endothelial cells; however, the downstream mediators have yet to be determined. Xu's team mapped the Gαq-R183Q phosphoproteome to identify pathogenic pathways and therapeutic targets.
To investigate Gαq-R183Q signaling in endothelial cells, they generated Gαq knockouts (KO) in immortalized human dermal microvascular endothelial cells (HDMECs) and rescued them with lentiviral expression of mTurquoise2-tagged Gαq-WT or Gαq-R183Q (Fig. 1A). Western blot confirmed similar Gαq expression in both rescued lines (~3-fold higher than parental HDMECs) (Fig. 1A, B). Immunofluorescence of VE-cadherin and F-actin showed that Gαq-R183Q induced cell elongation without affecting cell-cell junctions or cytoskeletal organization compared to Gαq-WT (Fig. 1C). Morphometric analysis confirmed significantly larger cell sizes in Gαq-R183Q HDMECs (Fig. 1D). Given that dysregulated endothelial migration and angiogenic sprouting drive vascular malformations, we investigated how Gαq-R183Q affects these functions. Scratch wound assays revealed that Gαq-R183Q significantly impaired HDMEC migration, resulting in delayed wound closure compared to Gαq-WT (Fig. 1E, F). In VEGF-induced spheroid-based sprouting assays, Gαq-R183Q strongly decreased sprout formation and elongation (Fig. 1G, H), confirming that proper Gαq signaling controls endothelial angiogenic behavior.
Ask a Question
Write your own review
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells
