Immortalized Human Cardiac Microvascular Endothelial Cells-SV40
Cat.No.: CSC-I9075L
Species: homo sapiens
Source: Cardiac Microvascular
Morphology: Polygonal
Culture Properties: Adherent
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Note: Never can cells be kept at -20 °C.
CIK-HT003 HT® Lenti-SV40T Immortalization Kit
free from contaminations (bacteria incl. mycoplasma, fungi, HIV, HAV, HBV, HCV, Parvo-B19) and cross-contaminations
Immortalized Human Cardiac Microvascular Endothelial Cells - SV40 are primary human cardiac microvascular endothelial cells (HCMVECs) that have been immortalized through the introduction of SV40 large T antigen, allowing for long-term and stable propagation. They maintain important characteristics of the cardiac microvascular endothelium while also providing increased practicality over primary endothelial cells due to extended lifespan and reduced donor variability, making them particularly well-suited for mechanistic studies and high-throughput screening.
The hallmark of these cells is their maintenance of a microvascular endothelial identity that is specific to the heart. They exhibit typical cobblestone morphology, establish tight cell-cell junctions, and express common endothelial markers such as CD31, VE-cadherin, von Willebrand factor (vWF), and endothelial nitric oxide synthase (eNOS). In terms of function, they respond strongly to shear stress, hypoxia, inflammatory cytokines, and angiogenic factors, which are all stimuli that are of particular relevance to the cardiac microenvironment. Thess cells have been used to study coronary microcirculation, endothelial dysfunction, and cardiac vascular inflammation. In general, they serve as a convenient and reliable in vitro model for the study of mechanisms underlying ischemia-reperfusion injury, diabetic cardiomyopathy, and heart failure-associated microvascular dysfunction.
Sufentanil Enhances the Viability and Inhibits the Apoptosis of OGD/R-Induced HCMECs
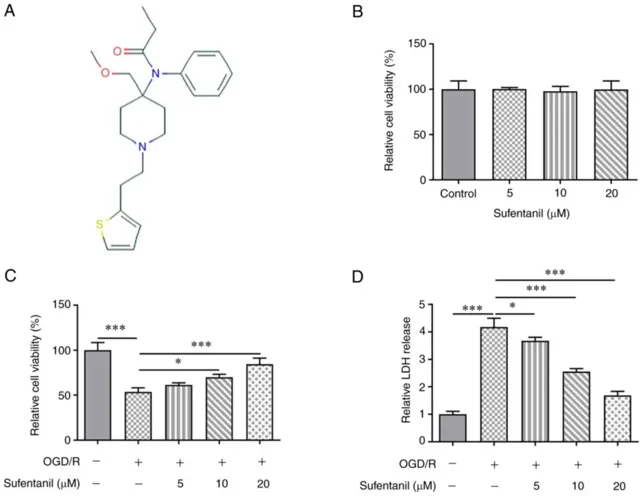
Ischemic heart disease is a major cause of death due to chronic myocardial damage from coronary artery ischemia. Here, Wang's team used oxygen and glucose deprivation/reoxygenation (OGD/R) to simulate myocardial I/R injury in HCMECs.
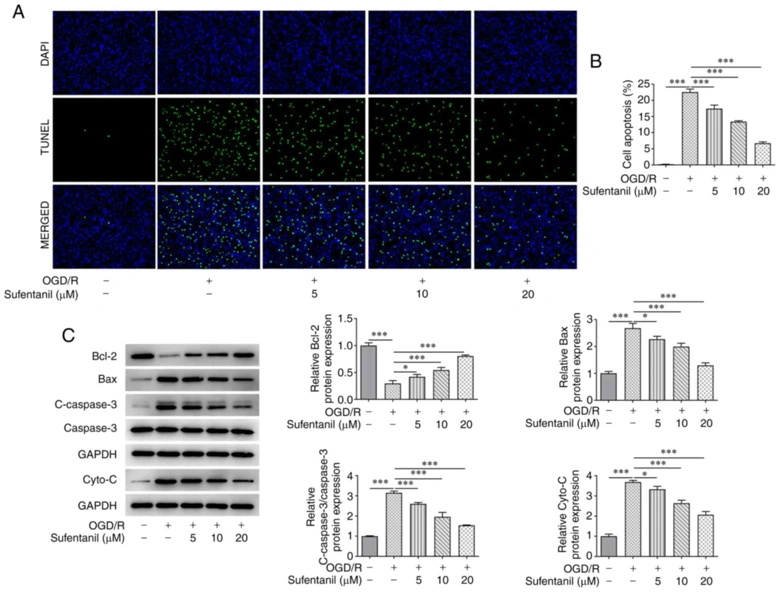
The chemical structure of sufentanil is shown in Fig. 1A. Sufentanil had no significant effect on HCMEC viability (Fig. 1B). OGD/R induction significantly decreased HCMEC viability, but sufentanil treatment partially enhanced it (Fig. 1C). Sufentanil also improved the viability of OGD/R-induced HCMECs in a dose-dependent manner. OGD/R induction increased LDH activity, which was gradually reduced by sufentanil treatment (Fig. 1D), indicating sufentanil's inhibitory effects on LDH activity. OGD/R induction significantly increased HCMEC apoptosis compared to controls, but sufentanil treatment suppressed this increase in a dose-dependent manner. 20 µM sufentanil had the best suppressive effect on apoptosis (Fig. 2A and B). OGD/R induction downregulated Bcl-2 and upregulated Bax, c-caspase-3, and cytochrome c protein expression. However, sufentanil treatment reversed these effects, as shown by upregulated Bcl-2 and downregulated Bax, c-caspase-3, and cytochrome c expression in the OGD/R + 5 µM, OGD/R + 10 µM, and OGD/R + 20 µM groups (Fig. 2C).
Ask a Question
Write your own review
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells
