Immortalized Human Breast Cancer Associated Fibroblasts-SV40
Cat.No.: CSC-I2088Z
Species: Human
Morphology: Polygonal
Culture Properties: Adherent
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
- Documents
Note: Never can cells be kept at -20°C.
Immortalized Human Breast Cancer-Associated Fibroblasts (BCAFs), established through the stable integration of the Simian Virus 40 (SV40) Large T antigen, constitute a sophisticated in vitro model for exploring the complex dynamics of the tumor microenvironment (TME). In breast cancer research, primary CAFs are critical mediators of tumor progression but are notoriously difficult to maintain due to early senescence and phenotypic drifting during ex vivo expansion. Our SV40-immortalized line overcomes these hurdles, providing a robust, high-fidelity platform for long-term mechanistic studies.
- Stable Activated Phenotype: These cells maintain the hallmark characteristics of activated myofibroblasts, including the persistent expression of Alpha-Smooth Muscle Actin (α-SMA) and Fibroblast Activation Protein (FAP). This ensures a consistent model for studying the stromal-driven metabolic reprogramming of cancer cells.
- Indefinite Proliferative Capacity: By bypassing the p53/pRb-mediated senescence checkpoints, this cell line offers an inexhaustible supply of standardized material. This scalability is essential for high-throughput screening (HTS) of novel compounds targeting the tumor stroma.
- Enhanced Secretome Fidelity: Our BCAFs reliably replicate the pro-tumorigenic secretome, including the release of specific growth factors (e.g., TGF-β, HGF) and pro-inflammatory cytokines (e.g., IL-6) that drive epithelial-mesenchymal transition (EMT) and chemoresistance in breast carcinoma cells.
- Optimized Co-culture Versatility: The line is engineered for high performance in 3D organoid models, transwell migration assays, and xenograft co-injection studies, providing a reproducible bridge between 2D cultures and complex in vivo stromal architecture.
Designed to replace the inherent variability of primary stromal cultures, the Immortalized Human BCAF-SV40 line provides a standardized, "assay-ready" system for pharmaceutical R&D. It is an indispensable tool for researchers aiming to decouple the intricate signaling pathways between the stroma and the tumor, ultimately accelerating the development of stroma-targeted therapies.
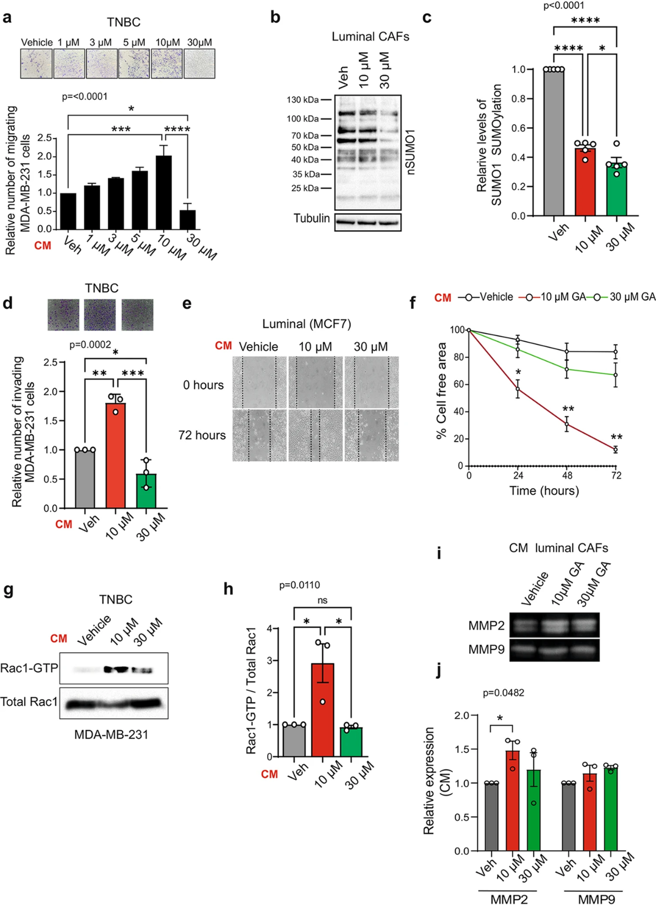
SUMOylation Regulates the Aggressiveness of Breast Cancer-Associated Fibroblasts
Cancer-associated fibroblasts (CAFs) are the most abundant stromal cellular component in the tumor microenvironment (TME). CAFs contribute to tumorigenesis and have been proposed as targets for anticancer therapies. Similarly, dysregulation of SUMO machinery components can disrupt the balance of SUMOylation, contributing to tumorigenesis and drug resistance in various cancers, including breast cancer. We explored the role of SUMOylation in breast CAFs and evaluated its potential as a therapeutic strategy in breast cancer.
We treated breast CAFs with the SUMO1 inhibitor ginkgolic acid (GA) at two different concentrations and conditioned media was used to analyze the proliferation, migration, and invasion of breast cancer cells from different molecular subtypes. Inhibition of SUMOylation with ginkgolic acid (GA) induces death in breast cancer cells but does not affect the viability of CAFs, indicating that CAFs are resistant to this therapy. While CAF viability is unaffected, CAF-conditioned media (CM) is altered by GA, impacting tumor cell behavior in different ways depending on the overall degree to which SUMO1-SUMOylated proteins are dysregulated. Breast cancer cell lines exhibited a concentration-dependent response to conditioned media (CM) from CAFs. At a low concentration of GA (10 µM), there was an increase in proliferation, migration and invasion of breast cancer cells. However, at a higher concentration of GA (30 µM), these processes were inhibited.
The study highlights the importance of SUMOylation in the tumor microenvironment, specifically in cancer-associated fibroblasts (CAFs).
Ask a Question
Write your own review
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells