Immortalized Human Bone Marrow-Derived Mesenchymal Stem Cells-SV40T
Cat.No.: CSC-I2037Z
Species: homo sapiens
Morphology: Polygonal
Culture Properties: Adherent
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Note: Never can cells be kept at -20°C.
Immortalized Human Bone Marrow-Derived Mesenchymal Stem Cells (hBM-MSCs), engineered through the stable transduction of the Simian Virus 40 Large T antigen (SV40T), represent a transformative tool for regenerative medicine and therapeutic development. While primary BM-MSCs are the "gold standard" in stem cell research, they are severely limited by donor-dependent variability and rapid in vitro senescence. Our SV40T-immortalized line effectively bypasses these constraints, providing a continuous, standardized, and highly proliferative cell population.
- Extended Proliferative Longevity: The integration of SV40T neutralizes the p53 and pRb pathways, ensuring indefinite expansion capabilities. This provides an inexhaustible cell source that eliminates the "culture fatigue" typical of primary MSCs, ensuring long-term experimental consistency.
- Maintenance of Multipotency: Despite their enhanced growth kinetics, these cells strictly retain their tri-lineage differentiation potential. They can be robustly induced into osteogenic, chondrogenic, and adipogenic lineages, making them ideal for tissue engineering and bone metabolism studies.
- Conserved Immunomodulatory Profile: Our hBM-MSCs maintain the hallmark low-immunogenicity (MHC Class I expression without MHC Class II) and characteristic surface markers (CD73+, CD90+, CD105+). They remain a high-fidelity model for investigating paracrine signaling and immunosuppressive mechanisms.
- Scalable Biopharmaceutical Platform: The line serves as a superior substrate for secretome analysis, exosome production, and viral vector manufacturing. Its standardized nature significantly reduces the technical noise associated with primary donor heterogeneity, accelerating the path from bench to clinical transition.
Engineered for precision and scalability, the Immortalized hBM-MSCs (SV40T) bridge the gap between basic stem cell biology and high-throughput industrial applications. This product is an essential asset for labs focusing on drug screening, gene therapy delivery, and the development of next-generation cell-based therapeutics.
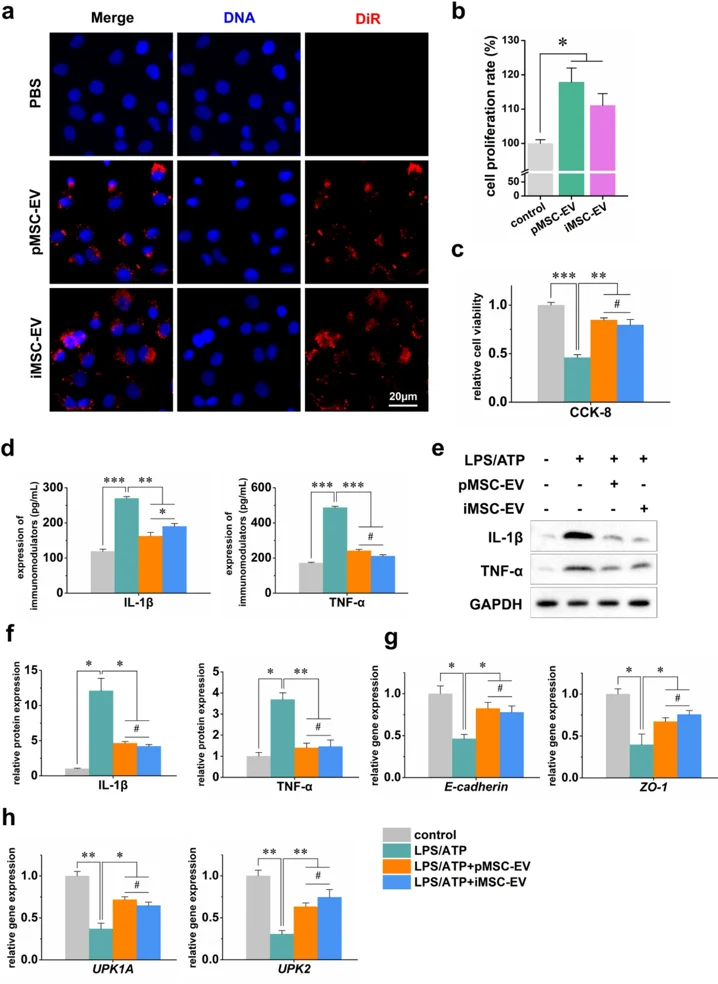
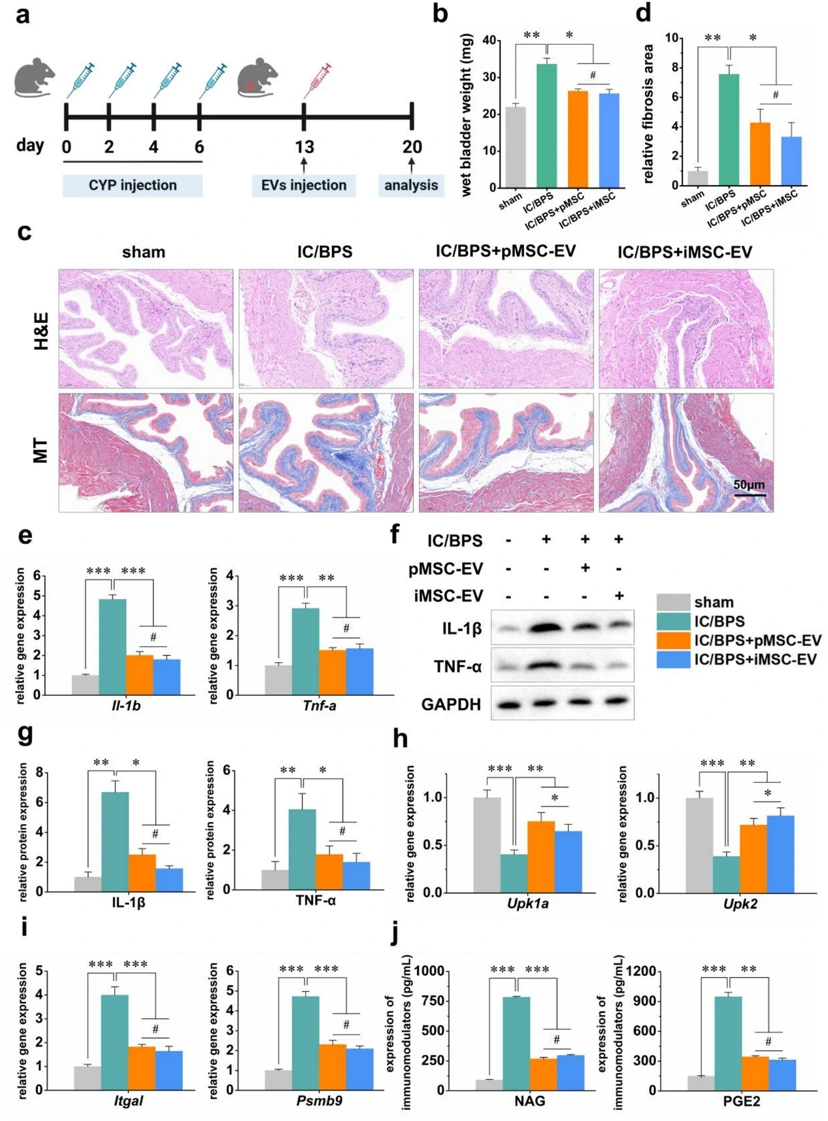
Immortalized Mesenchymal Stem Cell-Derived Extracellular Vesicles Represents a Promising Alternative to pMSCs for IC/BPS Therapy
Interstitial cystitis/bladder pain syndrome (IC/BPS) is a chronic condition characterized by debilitating pelvic pain. Mesenchymal stem cell-derived extracellular vesicles (MSC-EVs) are recognized as pivotal mediators of MSCs' paracrine activity and represent a novel therapeutic approach for IC/BPS. However, their efficacy is hindered by the inherent variability of primary MSCs (pMSCs) from different donors and their susceptibility to senescence during culture expansion. Immortalized mesenchymal stem cells (iMSCs) exhibited precisely controlled proliferation, maintained surface marker expression and differentiation capacities, comparable to pMSCs. We isolated EVs from iMSCs (iMSC-EVs) and compared their properties to those of EVs from pMSCs (pMSC-EVs). We further assessed the therapeutic effects of iMSC-EVs in both in vitro and in vivo models of IC/BPS. Our findings demonstrate that iMSC-EVs exhibit potent anti-inflammatory effects and confer protective benefits in IC/BPS.
Ask a Question
Write your own review
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells
