Immortalized Human Retinal Endothelial Cells-SV40T
Cat.No.: CSC-I2036Z
Species: homo sapiens
Morphology: Polygonal
Culture Properties: Adherent
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Note: Never can cells be kept at -20°C.
Immortalized Human Retinal Endothelial Cell line (iHRECs-SV40T) was developed from primary human retinal endothelial cells isolated and then immortalized by Simian Virus 40 Large T antigen (SV40T) insertion. The cell line has been used as an ophthalmic research model for BRB properties and vasodegenerative disorders.
Phenotypically, iHRECs-SV40T maintain crucial characteristics similar to primary cells. They express endothelial markers CD31 (PECAM-1), von Willebrand Factor (vWF), tight junction proteins ZO-1 and Occludin, which are responsible for the restrictive permeability characterizing the inner blood-retinal barrier. As they also show strong angiogenic properties in vitro, such as the capacity for tube formation, they are a widely used model for pathological angiogenesis, making them suitable for the study of diseases such as Diabetic Retinopathy (DR), Age-related Macular Degeneration (AMD) and Retinopathy of Prematurity (ROP). The cell line can be used to study the molecular mechanism of pathological neovascularization, inflammatory cytokine responses and oxidative stress in retinal microenvironment. They are also a useful research tool to better understand BRB physiology, while also providing a scalable platform for the evaluation of novel anti-angiogenic agents and delivery vehicles targeting the posterior segment of the eye in a model that is closer to human physiology than animal models.
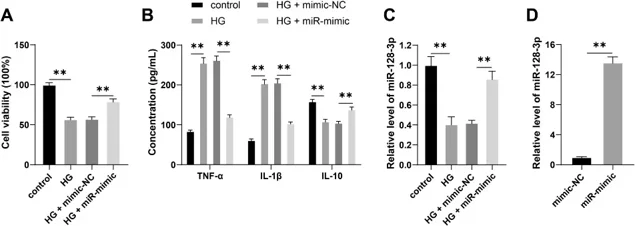
Overexpression of miR-128-3p Alleviates HG-induced DR Inflammatory Injury
Diabetic retinopathy (DR) is closely related to inflammatory injury. In this study, Wen et al. first established a DR cellular model by treating immortalized human retinal endothelial cells (IM-HRECs) with high-glucose (HG). They used the CCK-8 assay to detect cell viability, ELISA to measure the inflammatory cytokines (TNF-α, IL-1β, and IL-10), and RT-qPCR to detect the expression of miR-128-3p.
HG treatment significantly decreased cell viability and IL-10 expression (Fig. 1A, B) and markedly increased TNF-α and IL-1β levels (Fig. 1B) compared with the control group. This indicated that HG treatment induced DR inflammatory injury. RT-qPCR results also revealed that miR-128-3p expression was lower in the HG group than in the control group (Fig. 1C). This suggested that miR-128-3p might be a potential therapeutic target for DR. In order to verify the regulatory role of miR-128-3p in DR inflammatory injury, they increased miR-128-3p in IM-HRECs (Fig. 1C, D). Overexpression of miR-128-3p significantly improved the viability and IL-10 expression (Fig. 1A, B) and decreased TNF-α and IL-1β levels (Fig. 1B) in HG-treated cells. This indicated that overexpression of miR-128-3p could relieve HG-induced DR inflammatory injury.
Ask a Question
Write your own review
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells
