Immortalized Human Mammary Epithelial Cells (HMEC 2.6)
Cat.No.: CSC-I9116L
Species: Homo sapiens
Source: Mammary Tissue
Morphology: Polygonal
Culture Properties: Adherent
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Note: Never can cells be kept at -20 °C.
CIK-HT013 HT® Lenti-hTERT Immortalization Kit
CIK-HT003 HT® Lenti-SV40T Immortalization Kit
2) Soft agar assay preformed to determine anchorage-independence.
Immortalized Human Mammary Epithelial Cells 2.6 (HMEC 2.6) are epithelial cells isolated from normal mammary tissue. They have been genetically altered in a well-defined manner to allow for continuous growth in culture. These cells were immortalized with the intention of providing scientists with a mammary epithelial cell line that maintained characteristics of epithelial cells while allowing for greater longevity in culture than primary mammary epithelial cells. HMEC 2.6 cells characteristically show epithelial morphology and expression of epithelial markers such as cytokeratins and cell-cell adhesion proteins.
Due to their epithelial origin, HMEC 2.6 cells function similarly to normal breast epithelial cells. They can be used to study normal mammary gland biology including proliferation, differentiation, and epithelial cell interactions. When provided with the correct conditions, these cells will demonstrate organization that is characteristic of epithelial tissues. HMEC 2.6 cells also respond to growth factors and hormones in a manner similar to normal mammary epithelial cells. As these cells are derived from normal mammary tissue, they are often used as a control to compare the biology between normal breast cells and breast cancer cells.
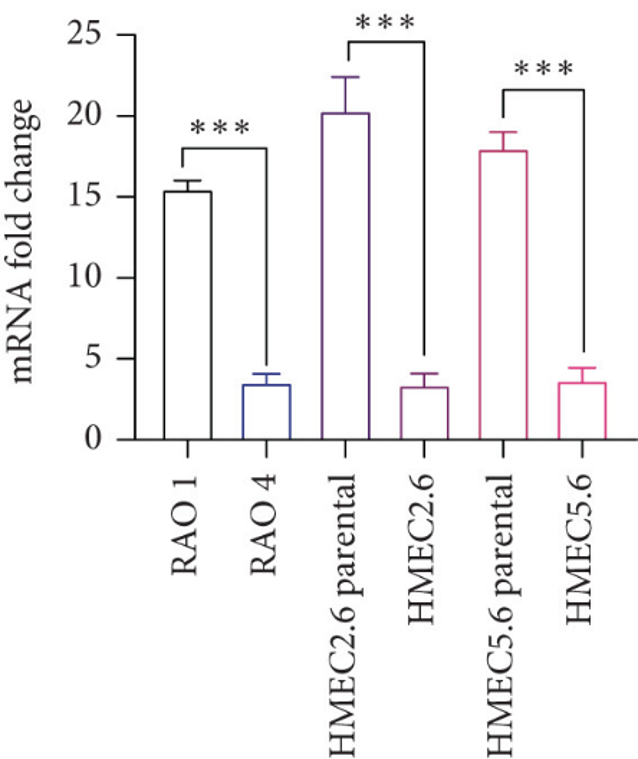
Reconfirmation of RAB25 Downregulation in HMEC Lines
RAB25 loss correlates with H-RAS mutations in triple-negative breast cancer (TNBC), but mechanisms driving tumor progression remain unclear. Joshi et al. wanted to further explore the cooperative effect of loss of RAB25 with activating mutations in H-RAS in HMECs. Previous studies have shown that loss of RAB25 acts as a tumor suppressor gene. However, it was unclear whether loss of RAB25 only cooperated with H-RAS61L specifically or with other oncogenes as well. To study this, immortal human mammary epithelial cell lines were created which had loss of RAB25. Cells named HMEC 2.6 and HMEC 5.6 were confirmed to have knockdown of RAB25 via shRNA. Quantitative PCR and western blot analysis were performed showing loss of RAB25 on both the mRNA and protein level when compared to parental controls (Fig. 1 and 2).

Ask a Question
Write your own review
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells
