Immortalized Human Hepatocytes-HPV
Cat.No.: CSC-I9339L
Species: Homo sapiens
Source: Liver
Culture Properties: Adherent
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Note: Never can cells be kept at -20 °C.
CIK-HT018 HT® Lenti-HPV-16 E6/E7 Immortalization Kit
The Immortalized Human Hepatocyte (IHH) cell line, established through the stable integration of Human Papillomavirus (HPV) E6 and E7 oncogenes, represents a pivotal advancement in liver-centric biotechnology. By neutralizing the p53 and pRb tumor suppressor pathways, this model bypasses the stringent Hayflick limit of primary human hepatocytes (PHHs), enabling sustained longitudinal studies without the loss of lineage-specific functionality.
- Preserved Metabolic Competence: Unlike traditional hepatoma-derived lines (e.g., HepG2 or Huh7) that exhibit significant metabolic derangement, HPV-immortalized hepatocytes maintain a high degree of differentiated function. They express critical Phase I and II drug-metabolizing enzymes, including key Cytochrome P450 (CYP) isoforms, facilitating more predictive toxicology and pharmacology assays.
- Superior Scalability & Reproducibility: These cells offer an inexhaustible supply for high-throughput screening (HTS). This eliminates the inter-donor variability inherent in PHH lots, providing researchers with a standardized, cost-effective baseline for global longitudinal data comparison.
- Enhanced Viral Susceptibility: The E6/E7-mediated immortalization maintains the expression of essential viral entry receptors (such as NTCP), rendering these cells an excellent substrate for studying Hepatitis B and C (HBV/HCV) replication kinetics and antiviral drug efficacy.
- Functional Synthetic Capacity: Our IHH-HPV line continues to synthesize and secrete essential plasma proteins, such as albumin and transferrin, and retains the ability for urea cycle activity, making it a robust model for metabolic disease research, including NAFLD and NASH.
Designed for the rigorous demands of pharmaceutical R&D, these cells serve as a high-fidelity surrogate for in vivo liver tissue. By offering a stable, biologically relevant, and "assay-ready" human cell system, the Immortalized Human Hepatocytes-HPV minimize the translational gap between in vitro discovery and clinical application, significantly accelerating the pipeline for novel hepatology therapeutics.
Cellular Senescence in Hepatocytes Contributes to Metabolic Disturbances in NASH
Cellular senescence is a state of irreversible cell cycle arrest and has been shown to play a key role in many diseases, including metabolic diseases. To investigate the potential contribution of hepatocyte cellular senescence to the metabolic derangements associated with non-alcoholic steatohepatitis (NASH), we treated human hepatocyte cell lines HepG2 and IHH with the senescence-inducing drugs nutlin-3a, doxorubicin and etoposide.
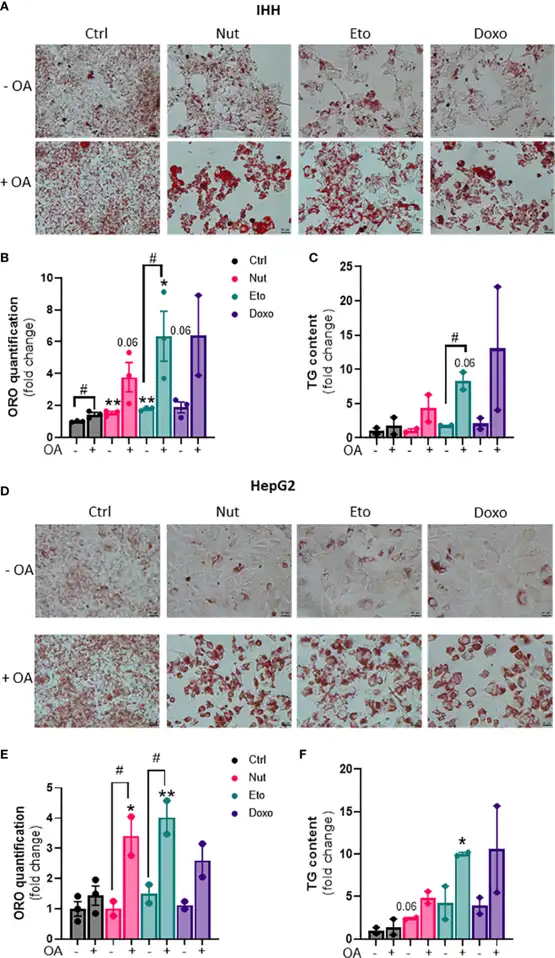
In order to determine whether cellular senescence affects hepatic lipid accumulation, lipid content was measured in senescent and control hepatocytes, in the absence or in the presence of oleic acid (OA) for 24h, in order to mimic pro-steatotic conditions. Hepatocytes treated with Nut, Eto and Doxo for 5 days displayed an increase in Oil Red O staining compared to control cells. In conditions with OA, the increased accumulation of lipids in senescent cells was even more pronounced (Fig. 1A, B, D, E). Quantification of intracellular triglycerides (TG) confirmed the increase in lipid content in senescent cells (Fig. 1C-F).
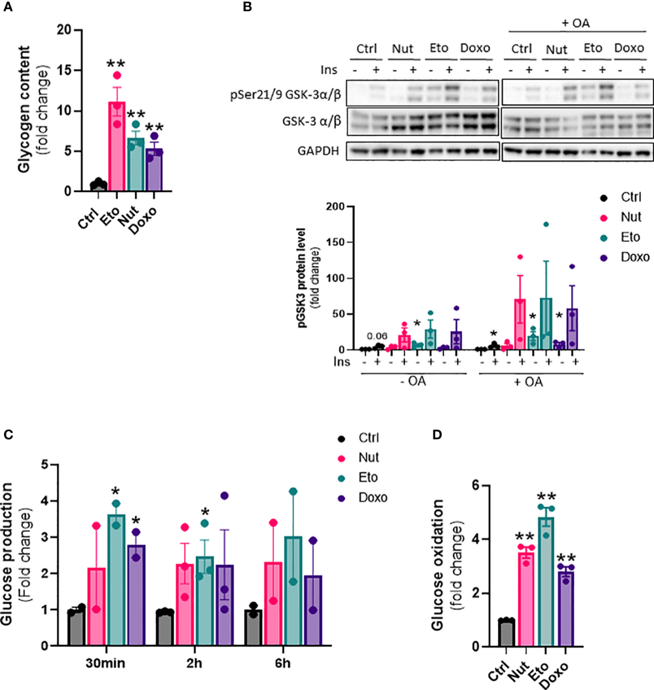
Next, we wanted to investigate the effect of cellular senescence on glucose metabolism. The results show that senescence causes changes in glucose metabolism in hepatocytes, including increased glycogen content (Fig. 2A), increased hepatic glucose output (Fig. 2C), as well as increased glucose oxidation capacity (Fig. 2D).
Together, these data indicate that hepatic senescence plays a causal role in the development of NASH pathogenesis, by modulating glucose and lipid metabolism, favoring steatosis.
Ask a Question
Write your own review
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells