Immortalized Human Conjuctival Epithelial Cells-SV40
Cat.No.: CSC-I9101L
Species: Homo sapiens
Source: Eye
Morphology: Polygonal
Culture Properties: Adherent
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Note: Never can cells be kept at -20 °C.
Generated from primary human conjunctival epithelium, immortalized human conjunctival epithelial cells-SV40 are able to proliferate indefinitely due to genetic modification induced by the introduction of the SV40 big T antigen. Bypassing replicative senescence, the immortalization process maintains epithelial polarity, mucin synthesis, and the ability to create a barrier, all of which are hallmarks of native conjunctival epithelial cells. Consequently, they offer a reliable and repeatable in vitro model that circumvents the short life span and inherent variability of original cultures. These cells are commonly utilized in ocular surface research to study epithelial physiology, inflammatory responses, and disease causes such as dry eye, allergic conjunctivitis, and infectious diseases. Research into cytokine signaling, cell-cell interactions, and the consequences of environmental stresses including oxidative damage and desiccation may all be accomplished using these assays. Furthermore, Immortalized Human Conjunctival Epithelial Cells-SV40 provide a useful platform for assessing ocular formulations, screening drugs for toxicity, and conducting drug screenings.
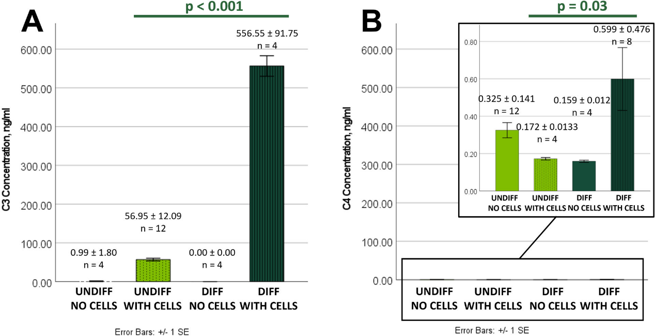
Immortalized Human Conjunctival Epithelial Cells Produce Functional Complement C3 and C4 Proteins
The complement system is crucial for ocular surface immunity, however it is uncertain whether conjunctival epithelial cells generate complement proteins. Ziemanski et al. investigated whether immortalized human conjunctival epithelial cells (HCjE) generate the complement proteins C3 and C4.
To assess C3 and C4 secretion by HCjE, supernatants from undifferentiated and differentiated cells were examined. C3 was identified in both circumstances but not in media controls, with differentiated HCjE secreting 9.77-fold more than undifferentiated cells (556.55 ± 91.75 vs. 56.95 ± 12.09 ng/ml, p < 0.001) (Fig. 1A). C4 levels were significantly reduced in all samples, with a peak in differentiated HCjE supernatants (0.599 ± 0.476 ng/ml) (Fig. 1B). The cell lysate analysis corroborated these findings. Differentiated HCjE had 3.25 times more cellular C3 than undifferentiated cells (0.585 ± 0.183 vs. 0.180 ± 0.102 ng/μg protein, p < 0.001) (Fig. 2A). In contrast, cellular C4 was 3.95-fold lower in differentiated vs undifferentiated lysates (0.00202 ± 0.00016 vs. 0.00797 ± 0.00487 ng/μg protein, p < 0.001) (Fig. 2B).
![C3 and C4 from HCjE Cell Lysates HCjE were cultured under undifferentiating (UNDIFF, serum-free) and differentiating (DIFF, 10% heat-inactivated serum) conditions (see Methods) and used to prepare HCjE cell lysates for analysis of C3 and C4 fractions, defined to be total [C3 or C4] per ug of total cellular protein](/upload/images/immortalized-human-conjuctival-epithelial-cells-sv40-casestudy-2.webp)
Ask a Question
Write your own review
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells