Mouse Hepatic Stellate Cells
Cat.No.: CSC-C4736Z
Species: Mouse
Source: Liver
Cell Type: Hepatic Stellate Cell
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Never can cryopreserved cells be kept at -20 °C
Mouse Hepatic Stellate Cells (MHSCs) are mouse non-parenchymal liver cells situated in the space of Disse, where they are found between hepatocytes and sinusoidal endothelial cells. Hepatic stellate cells are at the center of many aspects of liver physiology, regeneration, and pathology including fibrosis. Quiescent hepatic stellate cells store vitamin A (retinoids) as cytoplasmic lipid droplets and modulate extracellular matrix homeostasis.
Quiescent stellate cells are morphologically distinct because of their star shape and numerous cytoplasmic processes. When liver injury or profibrotic mediators (TGF-β, PDGF) are present hepatic stellate cells activate into myofibroblast-like cells. Activated hepatic stellate cells begin to proliferate, lose their retinoid droplets, express higher levels of α-smooth muscle actin (α-SMA), and secrete large amounts of ECM components like collagen I and III, fibronectin, and laminin.
In vitro, Mouse Hepatic Stellate Cells are cultured as adherent fibroblast-like cells and are widely used for studies of fibrogenesis, liver injury, inflammation, and hepatotoxicity. They provide a platform to investigate the molecular mechanisms underlying stellate cell activation, ECM deposition, oxidative stress, cytokine signaling, and cell-cell interactions within the hepatic microenvironment.
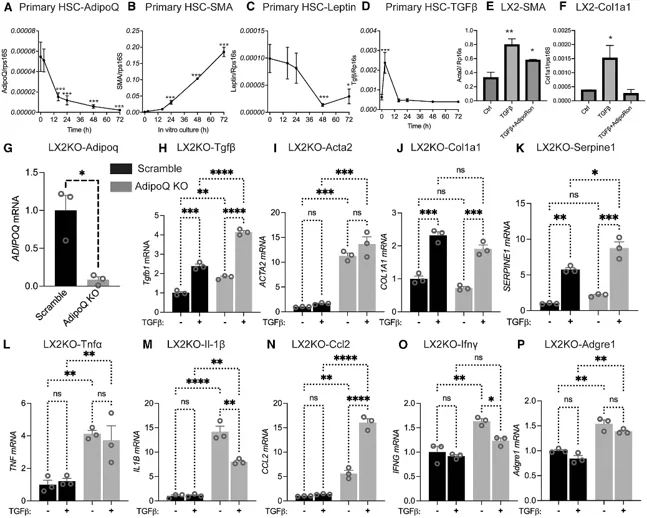
The Expression of Adiponectin and Fibrotic Genes is Inversely Correlated in HSCs
Hepatic stellate cells (HSCs) drive liver fibrosis. Although adiponectin is expressed in quiescent HSCs, its local role in fibrosis progression remains unclear. Zhao's team investigated whether HSC-specific adiponectin regulates fibrosis development using an inducible transgenic mouse model.
Activated hepatic stellate cells (HSCs) are the primary source of collagen and responsible for liver fibrosis. To determine if adiponectin produced locally by HSCs regulated HSC activation, wild-type mouse HSCs were isolated and gene expression was analyzed over time. Adiponectin expression decreased rapidly within 2 hours of culture, prior to induction of fibrotic marker SMA (Fig. 1A, 1B), indicating adiponectin may inhibit expression of fibrotic genes. Leptin expression also decreased, but at a slower rate (Fig. 1C). TGF-β expression peaked initially at 2 hours then returned to baseline levels (Fig. 1D), which may be a result of activation from cell isolation. Next, they treated human LX2 HSCs with TGF-β and found increased expression of fibrotic markers SMA and COL1A1 that were reversed by treatment with adiponectin receptor agonist AdipoRon (Fig. 1E, 1F). Adiponectin knockdown in LX2 cells using CRISPR-Cas9 (>90% reduction; Fig. 1G) significantly increased baseline and TGF-β-induced expression of Tgfβ, Acta2, Col1a1, and Serpine1 (Fig. 1H-1K). Adiponectin deletion alone was sufficient to induce increases in Tgfβ and Acta2 expression. Knockdown of adiponectin also significantly induced expression of inflammatory genes TNFα, IL-1β, Ccl2, Ifnγ, and Adgre1 even in the absence of TGF-β (Fig. 1L-1P).
Ask a Question
Write your own review