Immortalized Rat Retinal Capillary Endothelial Cells-SV40
Cat.No.: CSC-I9347L
Species: Rattus norvegicus
Source: Eye
Morphology: Polygonal
Culture Properties: Adherent
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Note: Never can cells be kept at -20 °C.
Immortalized Rat Retinal Capillary Endothelial cell line is a primary cell line with a high degree of specialization, as it was derived from retinal capillaries of transgenic rats and immortalized by the integration of Simian Virus 40 (SV40) large T-antigen, under a temperature-sensitive promoter or transduced by the virus. Thus, these cells escape senescence seen in primary cells, and enable the study of an in vivo endothelium of the inner Blood-Retinal Barrier (iBRB) in a more reproducible and long-term manner.
The immortalized retinal capillary endothelial cells share most of the key characteristics seen in the retinal endothelium. This includes a polarized monolayer with high electrical resistance and low paracellular permeability, due to the expression of tight junction proteins, Claudin-5, Occludin and ZO-1. They also express several nutrient transporters, GLUT1 for glucose and several amino acid transporters, essential for supplying nutrients to neural retina. The cell line is often employed in pharmacological and pathological studies that need to test for leukocyte adhesion, oxidative stress, and angiogenic factors, under conditions of retinal disease. These cells are used primarily as an in vitro model of blood-retinal barrier breakdown in Diabetic Retinopathy (DR), and to screen transport kinetics of ophthalmic drugs across vascular wall.
The Anti-Inflammatory Effects of L-DOPA/DA in EAU Gnat1rd17 Mice Are Mediated by the Suppression of NF-κB (p65) and STAT3 Activity in Retinal Endothelial Cells
The gateway reflex regulates immune cell entry into tissues via neural inputs. Here, Stofkova et al. hypothesized that rod photoreceptor dysfunction in Gnat1rd17 mice remodels retinal neural activity, influencing the blood-retinal barrier and EAU development.
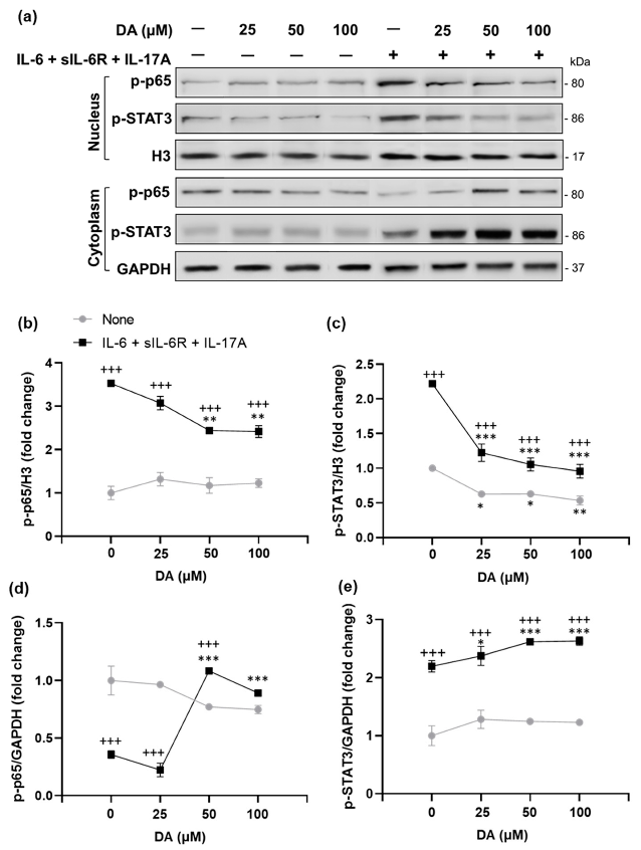
They used transcriptome profiling, target identification, and functional validation to investigate retinal remodeling and its effects on EAU. Results showed that Gnat1rd17 mice exhibited altered retinal dopaminergic systems, leading to exacerbated EAU, which was mitigated by dopamine replacement with L-DOPA. Then, they explored the impact of DA on pro-inflammatory cytokine (IL-6, sIL-6R, and IL-17A)-induced p-p65 and p-STAT3 expression in rat retinal capillary endothelial cells. DA pretreatment significantly decreased nuclear p-p65 and p-STAT3 levels in a dose-dependent manner (Fig. 1a-c), with increased cytoplasmic expression (Fig. 1a, d, e). DA also reduced nuclear p-STAT3 expression under basal conditions, without cytokine stimulation (Fig. 1a, c). Since NF-κB and STAT3 activate pro-inflammatory genes that recruit CD4+ T cells in the retina, we evaluated mRNA expression of IL-6 and T-cell-attracting chemokines (Ccl20, Ccl2, and Cxcl10) under basal and cytokine-stimulated conditions in DA-pretreated cells. DA reduced mRNA expression of IL-6 and all measured chemokines in cytokine-stimulated cells (Fig. 1f). Under basal conditions, DA pretreatment trended towards inhibiting IL-6 and chemokine expression, with significant suppression of Ccl20 and Cxcl10 (Fig. 1f). Overall, these findings demonstrate that L-DOPA/DA mitigates EAU by reducing NF-κB (p65) and STAT3 activation and expression of pro-inflammatory target genes in retinal endothelial cells, thereby protecting the BRB from inflammatory damage.
Ask a Question
Write your own review
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells