Immortalized Mouse Olfactory Bulb Cells (OBC1)
Cat.No.: CSC-I9285L
Species: Mus musculus
Source: Olfactory bulbs
Morphology: Flat, process-bearing cells
Culture Properties: Adherent
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Note: Never can cells be kept at -20 °C.
2) Southern or Northern blot analysis of the viral integration site
3) immunocytochemical analysis for markers.
The Immortalized Human Retinal Endothelial Cell line (iHRECs-SV40T) is an immortalized cell line of primary human retinal endothelial cells (HRECs) that has been immortalized with the Simian Virus 40 Large T antigen (SV40T). The immortalized endothelial cells maintain an appropriate phenotype after long term passages and can be used for high-throughput screening and extended experiments.
These cells physiologically resemble primary HRECs. The cell line expresses CD31 (PECAM-1), von Willebrand Factor (vWF), ZO-1, Occludin, and many other endothelial markers. These markers are used as an endothelial marker in cell culture and maintain the restrictive permeability of the inner blood-retinal barrier (BRB). Due to the high angiogenic potential of these cells, their in vitro ability to form tubes, and as an in vivo cause of many vasodegenerative disorders, they are often used to study Diabetic Retinopathy (DR), Age related Macular Degeneration (AMD), and Retinopathy of Prematurity (ROP).
The iHRECs-SV40T cell line can be used to model molecular pathways of pathological neovascularization, inflammatory cytokines and oxidative stress in the retinal microenvironment. This cell line helps to facilitate new therapeutic development between animal models and clinical use as it is a scalable and human derived model for testing of new anti-angiogenic drugs and delivery systems to the posterior segment of the eye.
Ammonia Toxicity Aggravates Mitochondrial Dysfunction, Cell Death, ROS Generation, and the Secretion of Pro-Inflammatory Cytokines in OBC1 Olfactory Bulb Cells
Hepatic encephalopathy (HE) is a neuropsychiatric disorder associated with cirrhosis and chronic liver disease primarily driven by ammonia (NH3) toxicity, which leads to neuroinflammation and cognitive deficits. Recent studies have identified olfactory dysfunction as a potential early indicator of HE, linked to ammonia-induced neurotoxicity in the brain.
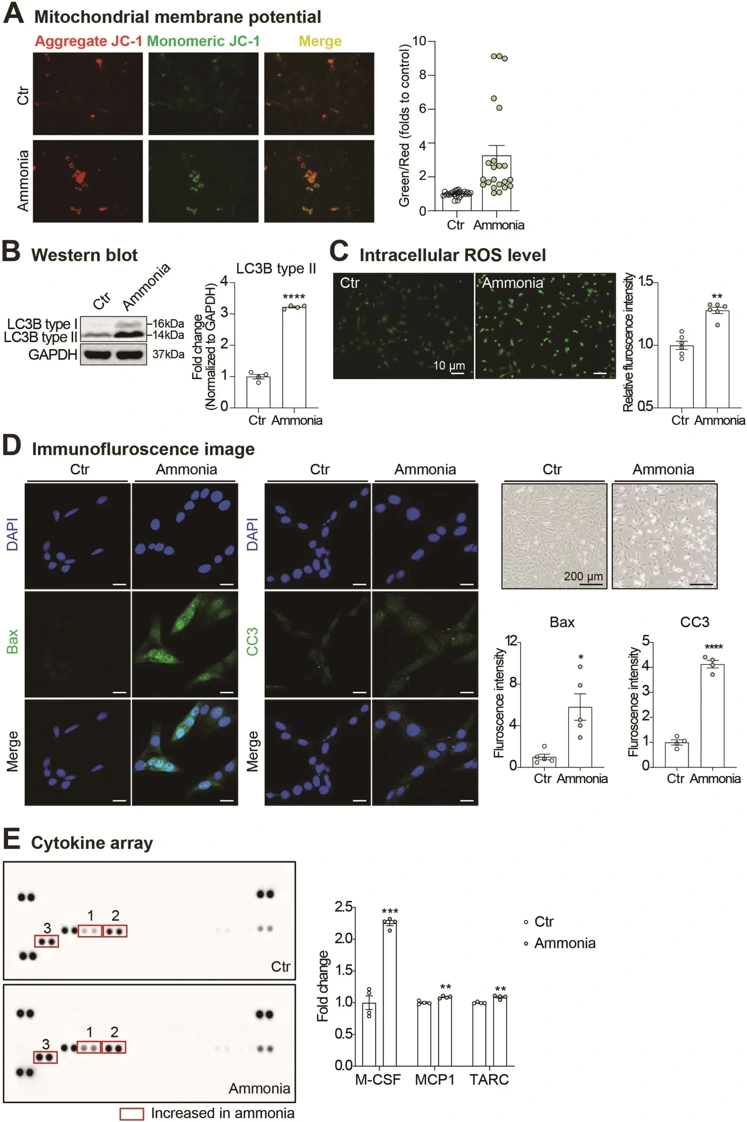
To gain a better understanding of the effect of ammonia toxicity on olfactory bulb cells, Kim et al. performed a series of experiments on the OBC1 cells immortalized mouse olfactory bulb cell line. The results of the study indicated that ammonia markedly impaired mitochondrial function, demonstrated by loss of membrane potential (Fig. 1A). The results also demonstrated that autophagy activity was increased following ammonia treatment, which was confirmed by a significant increase in LC3B type II expression (Fig. 1B). The presence of oxidative stress was confirmed by an increase in intracellular ROS levels visualized by DCF-DA fluorescence (Fig. 1C). The study also demonstrated that ammonia significantly caused cell death in OBC1 cells, as shown by an increase in the expression of CC3 and Bax (Fig. 1D). In the inflammation experiment, ammonia was shown to increase the secretion of pro-inflammatory cytokines M-CSF, MCP1, and TARC (Fig. 1E). In summary, the results of this study demonstrate that increased ammonia levels as seen in hepatic encephalopathy (HE) may cause significant cellular dysfunction in olfactory bulb cells, which could be the potential cause of olfactory dysfunction in HE patients.
Ask a Question
Write your own review
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells