Immortalized Mouse Hepatocytes-SV40T
Cat.No.: CSC-I2101Z
Morphology: Polygonal
Culture Properties: Adherent
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Note: Never can cells be kept at -20°C.
Immortalized Mouse Hepatocytes-SV40T, derived from mice, are hepatocytes that have been genetically modified to constitutively express the SV40 large T antigen. The expression of SV40T allows them to circumvent replicative senescence and proliferate indefinitely in vitro.
Immortalized Mouse Hepatocytes-SV40T are polygonal epithelial cells that can be cultured as monolayer adherent cells. They express albumin (Alb), AFP, CK18, HNF-1α, HNF-4α, and sometimes will express functional phase I (CYP450 family members) and phase II (UGTs, GSTs) metabolism enzymes if maintained appropriately. They may also store glycogen, produce urea and respond to certain hormones such as insulin or glucagon. They are commonly utilized for investigations into liver metabolism, metabolism of xenobiotics, hepatitis virus infection, lipid metabolism, liver regeneration, transformation and cytotoxicity studies. They are also utilized for transfection experiments and CRISPR knockout studies.
ER Stress Upregulates S100A11 in Steatohepatitis Models Via Epigenetic Modifications Within the Lipotoxicity-Influenced Enhancer
The pathogenesis of metabolic dysfunction-associated steatohepatitis (MASH) involves lipotoxicity, endoplasmic reticulum (ER) stress, and inflammation. Daniel et al. show that lipotoxicity-induced ER stress epigenetically upregulates the proinflammatory alarmin S100A11 and illuminates how to therapeutically target S100A11.
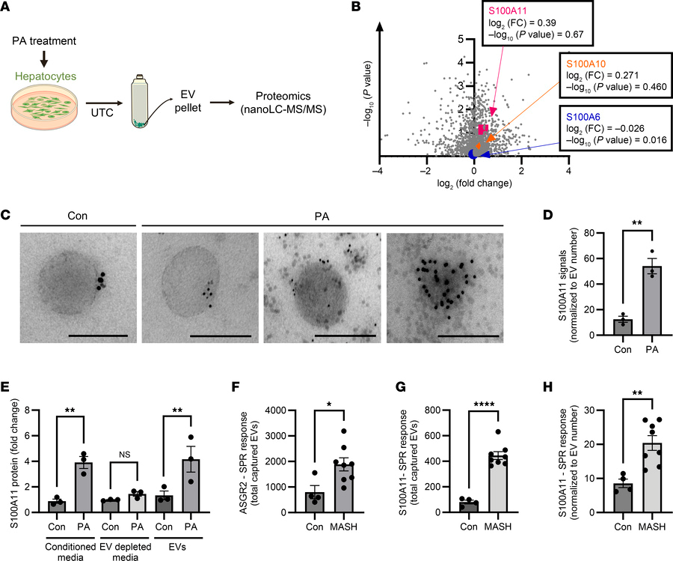
To determine which DAMPs are enriched in EVs shed by lipotoxic hepatocytes, the authors performed proteomics on EVs secreted by palmitate-treated immortalized mouse hepatocytes (WT-IMH). Bioinformatic processing identified 1660 proteins with altered expression, including 44 DAMPs (Fig. 1A). Of these, S100A11 was the most significantly upregulated protein in palmitate-treated WT-IMH EVs (Fig. 1B). Validation of S100A11 enrichment in EVs was performed using multiple methods. Immunogold EM demonstrated increased cell surface expression of S100A11 on EVs derived from palmitate-treated primary mouse hepatocytes (Fig. 1C). SPR quantification using anti-ASGR1 antibody-captured EVs confirmed S100A11 enrichment on palmitate-stimulated EVs (Fig. 1D). In human Huh7 cells, Western blotting showed increased S100A11 in EV lysates from palmitate-treated cells. ELISA of conditioned supernatants revealed higher S100A11 with palmitate treatment, which disappeared upon EV depletion; isolated EVs confirmed S100A11 enrichment (Fig. 1E). Thus, palmitate-induced lipotoxicity drives release of S100A11-enriched EVs.
Ask a Question
Write your own review
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells