COR-L23
Cat.No.: CSC-6253W
Species: Homo sapiens (Human)
Source: Pleural Effusion Metastasis
Morphology: continuous culture, grown as monolayer, morphology epithelial
Culture Properties: monolayer
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Tissue: lung;
Tumor: carcinoma, large cell;
Derived from: pleural effusion
COR-L23 is an epithelial cell line derived from human lung carcinoma. It was initially developed from the pleural effusion of a patient suffering from non-small cell lung cancer (NSCLC). COR-L23 is a metastatic-derived cell line that has been used as an in vitro lung tumor model for studying cancer cell dissemination and response to therapeutics.
The cells have been reported to predominantly form adherent epithelial-like monolayers when cultured and they possess a moderately high doubling time when maintained in routine cell culture conditions. Analysis of the karyotype is reported to be aneuploid, which is consistent with other lung carcinomas. Additionally, at the molecular level the COR-L23 cell line has dysregulated pathways controlling the cell cycle, apoptosis, and DNA damage. As such, COR-L23 can be used to further delineate the mechanisms of tumor proliferation and survival. COR-L23 has been used to study chemotherapeutic drugs as well as radiation and combination therapies as a model for lung cancer. The cells have also been used to study drug resistance mechanisms and discover potential biomarkers for NSCLC.
Differences in Activity and Expression of STAT3 and Everolimus-Induced Expression Changes of EMT Indicators in Lung-Derived Cell Lines
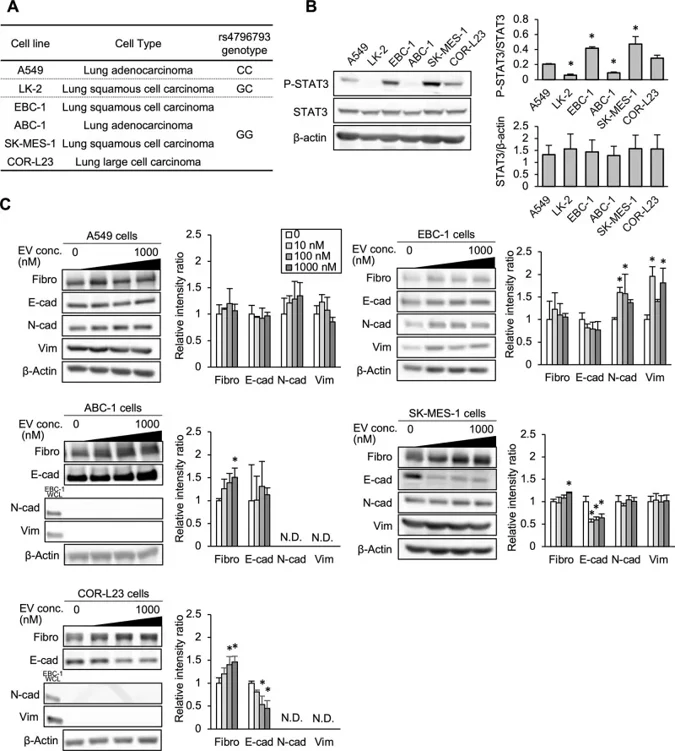
Interstitial lung disease (ILD), a potentially fatal and dose-limiting toxicity, is associated with mTOR inhibitors in metastatic renal-cell carcinoma (RCC). Yamamoto et al. investigated whether STAT3 polymorphism rs4796793 (-1697C/G) influences risk of ILD and explored potential mechanisms using lung-derived cell lines.
Six lung-derived cell lines possessed unique genotypes (A549: CC, LK-2: GC, all other cell lines: GG; Fig. 1A). STAT3 expression levels were independent of genotype, although STAT3 activation (p-STAT3/total STAT3) was higher in EBC-1 and SK-MES-1 cells than in A549 cells, and no consistent pattern was observed among the lines based on genotype. Everolimus altered EMT marker expression in a cell-line dependent manner (Fig. 1C). In EBC-1, it significantly upregulated mesenchymal indicators N-cadherin and vimentin; in ABC-1, SK-MES-1, and COR-L23, it increased fibronectin; and in SK-MES-1 and COR-L23, it decreased epithelial marker E-cadherin. Notably, A549 cells showed no significant EMT changes at equivalent drug concentrations. N-cadherin and vimentin were undetectable in ABC-1 and COR-L23 by Western blot.
Ask a Question
Write your own review
- You May Also Need
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells