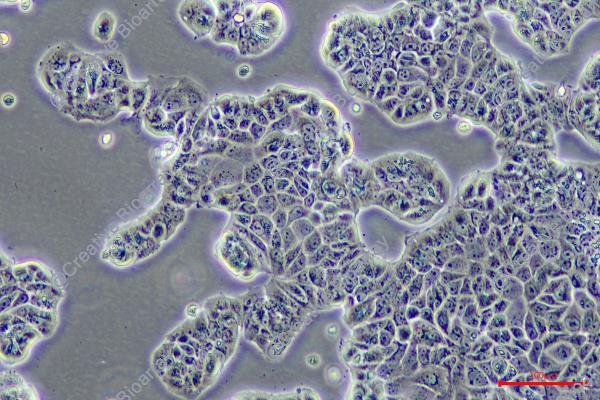
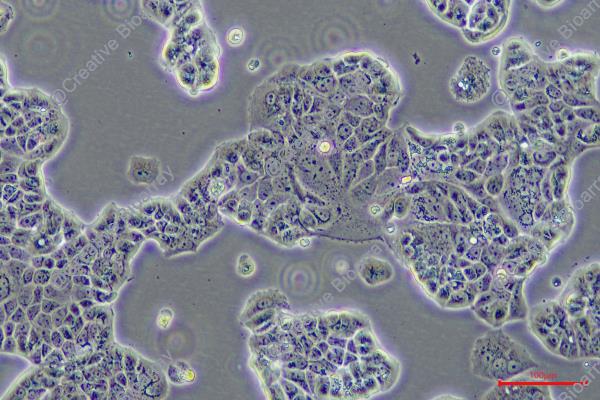
EFM-192A
Cat.No.: CSC-C0358
Species: Homo sapiens (Human)
Source: Pleural Effusion Metastasis
Morphology: epitheloid, adherent growing as monolayer
Culture Properties: monolayer
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Immunology: cytokeratin +, cytokeratin-7 +, cytokeratin-8 +, cytokeratin-17 -, cytokeratin-18 +, cytokeratin-19 +, desmin -, endothel -, EpCAM +, GFAP -, neurofilament -,
The EFM-192A cell line is a well-characterized human breast carcinoma model established from the malignant pleural effusion of a patient with metastatic breast cancer. It is classified as a HER2-positive (ERBB2-amplified) breast cancer model, representing a clinically significant subtype that accounts for approximately 15-20% of breast cancers and is associated with aggressive disease. Beyond HER2 amplification, EFM-192A harbors a second key driver mutation: an activating mutation in the PIK3CA gene (H1047R), which is one of the most frequent oncogenic alterations in breast cancer. This co-occurrence of HER2 amplification and PIK3CA mutation makes EFM-192A a genetically representative model of a common and therapeutically challenging tumor profile, reflecting the complex genomic landscape often seen in clinical practice.
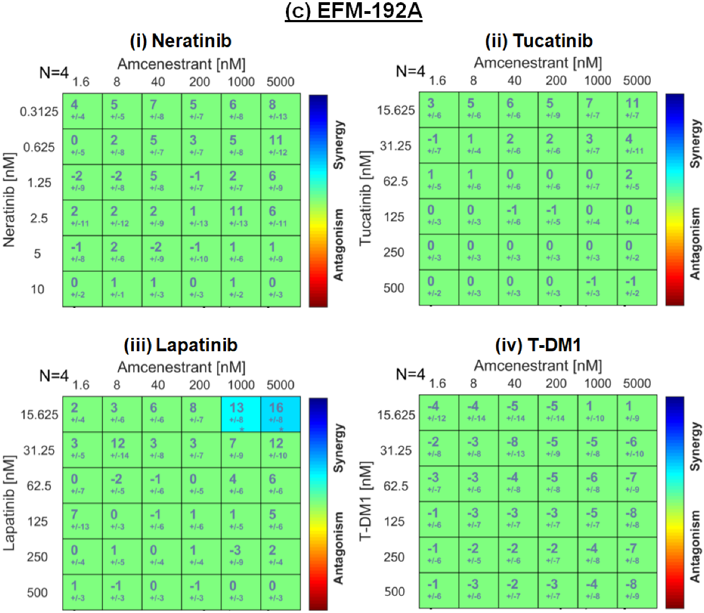
HER2-Targeted Agents and Amcenestrant Are Synergistic in HER2+/ER+ Cell Lines
HER2-positive/oestrogen receptor-positive (HER2+/ER+) represents a unique breast cancer subtype. The use of individual HER2- or ER-targeting agents can lead to the acquisition of therapeutic resistance due to compensatory receptor crosstalk. New drug combinations targeting HER2 and ER could improve outcomes for patients with HER2+/ER+ breast cancer. In this study, the pre-clinical rationale is explored for combining amcenestrant (Amc), a selective oestrogen receptor degrader (SERD), with HER2-targeted therapies including trastuzumab, trastuzumab-emtansine (T-DM1) and tyrosine kinase inhibitors (TKIs). The combination of Amc and anti-HER2 therapies was investigated in a panel of four HER2+/ER+ cell lines: BT-474, MDA-MB-361, EFM-192a and a trastuzumab-resistant variant BT-474-T.
Additivity and synergy were observed between Amc and the TKIs neratinib, lapatinib and tucatinib in all cell lines. TKI/Amc combinations reduced p-HER2 and ER levels and resulted in increased apoptosis. In conclusion, the combination of Amc- and HER2-targeted treatments has potential as a therapeutic strategy for the treatment of HER2+/ER+ breast cancer and warrants further clinical investigation to validate safety and efficacy in patients.
Ask a Question
Write your own review
- You May Also Need
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells