RKO-E6
Cat.No.: CSC-C9244W
Species: Homo sapiens (Human)
Source: Intestine; Colon
Morphology: epithelial
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
RKO-E6 Cells (RKO-hE6) is a cell line derived from the human colon adenocarcinoma cell line RKO that has been engineered to constitutively express the E6 oncoprotein from human papillomavirus. E6 expression leads to degradation of the tumor suppressor protein p53 and thus renders RKO-E6 cells p53-null. Comparing these cells with the p53-competent parental RKO line creates a perfect system for exploring p53's role in cancer research.
Like their parental cell line RKO, these cells maintain an adherent epithelial growth pattern when maintained in tissue culture. Although these cells closely mimic parental RKO cells, the lack of p53 significantly alters cell cycle checkpoints, response to DNA damage, apoptosis, and maintenance of genomic integrity. Cells lacking p53 are often resistant to DNA-damaging agents and show altered transcriptional regulation when compared to p53-competent cells. As such, these cells are often used to study tumor suppressors, cancer cell survival, and chemoresistance.
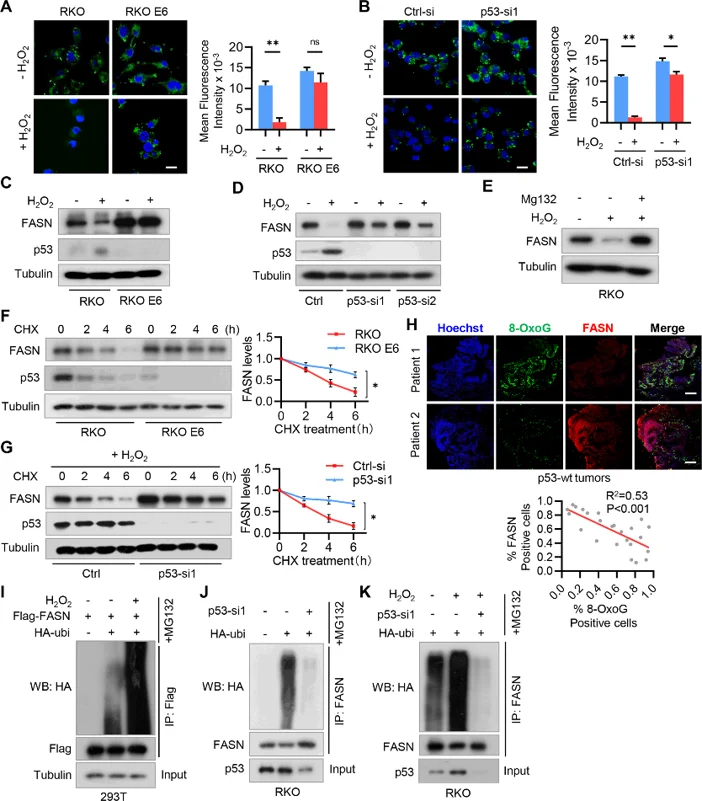
H2O2 Inhibits Fatty Acid Accumulation by Promoting FASN Ubiquitination and Degradation in P53+/+ CRC Cells
Oxidative stress and altered lipid metabolism are two characteristics of cancer cells. Han et al. investigated the crosstalk between these two biological processes in CRC.
Oxidative stress agent H₂O₂ acutely reduced lipid droplets in p53 wild-type CRC cells (RKO), but not p53-null CRC cells (RKO E6). Lipid accumulation inhibition by H₂O₂ was compromised by p53 knockdown, suggesting that p53 mediates fatty acid accumulation suppression in CRC cells (Fig. 1A, B). Additionally, H₂O₂ treatment led to induction of p53 and inhibition of FASN protein expression without altering their mRNA levels in RKO cells (Fig. 1C, D). FASN repression by H₂O₂ was rescued by p53 knockdown (Fig. 1D). Proteasome inhibitor MG132 abrogated H₂O₂-mediated FASN repression (Fig. 1E). FASN protein is more stable in p53-null CRC cells compared with p53-wild-type CRC cells, and p53 knockdown suppressed FASN protein degradation (Fig. 1F, G). Furthermore, FASN expression inversely correlated with oxidative DNA damage marker 8-OxoG in p53 wild-type but not null CRC tissues (Fig. 1H). Finally, H₂O₂ increased FASN ubiquitination in a p53-dependent manner (Fig. 1I-K). Taken together, these findings reveal that FASN is destabilized by H₂O₂ via p53-dependent ubiquitination in CRC.
Ask a Question
Write your own review
- You May Also Need
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells