RH-18
Cat.No.: CSC-C0502
Species: Homo sapiens (Human)
Source: Perineus; Muscle
Morphology: large transparent epitheloid cells, partially with long extensions, growing as monolayers
Culture Properties: monolayer
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Immunology: cytokeratin -, desmin +, endothel -, EpCAM -, GFAP -, neurofilament -, vimentin +
Viruses: PCR: EBV -, HBV -, HCV -, HIV -, HTLV-I/II -, SMRV -
RH-18 is a human rhabdomyosarcoma (RMS) cell line established from a primary pediatric embryonal tumor serially xenografted in immunodeficient mice. Its paramount scientific advantage lies in its molecular classification: RH-18 represents a fusion-negative alveolar RMS (aRMS), lacking the canonical PAX3/FOXO1 translocation. This positions it as a superior and physiologically relevant model for dissecting the heterogeneous, often fusion-negative, RMS subtypes that dominate clinical presentation yet are underrepresented by traditional cell lines.
Originating from the St. Jude Children's Research Hospital xenograft program (SJRH-18), the line is authenticated via STR profiling and retains a complex hypotetraploid karyotype mirroring its primary tumor origin. Functionally, RH-18 maintains a mesenchymal/myogenic identity (desmin+, vimentin+, fetal muscle antigen 5.1H11+, creatine phosphokinase+), ensuring phenotypic relevance. With a moderate doubling time (~60-70 h) and negative virology (EBV-, HBV-, HCV-, HIV-, HTLV-), it offers a robust, biosafe platform for high-throughput screening (ChIP-seq, RNA-seq) and preclinical therapeutic testing. RH-18 thus serves as an indispensable reference standard for investigating non-translocation-driven RMS oncogenesis and therapeutic resistance.
Pannexin 1 Induces Rhabdomyosarcoma Cell Fusion by Downregulating APOBEC2
Rhabdomyosarcoma (RMS) is an aggressive cancer thought to arise from impaired myogenesis. This can be substantially overcome by increasing the levels of pannexin 1 (PANX1), a critical component of the myogenic program, but the mechanism involved is unknown.
Using RNA-seq, we have previously found that overexpression of PANX1 dramatically reshapes the transcriptomic landscape of RMS including downregulation of a myogenic modulator, APOBEC2 (apolipoprotein B mRNA editing enzyme catalytic subunit 2). Following this clue, we investigated the regulation of APOBEC2 expression by PANX1 in RMS.
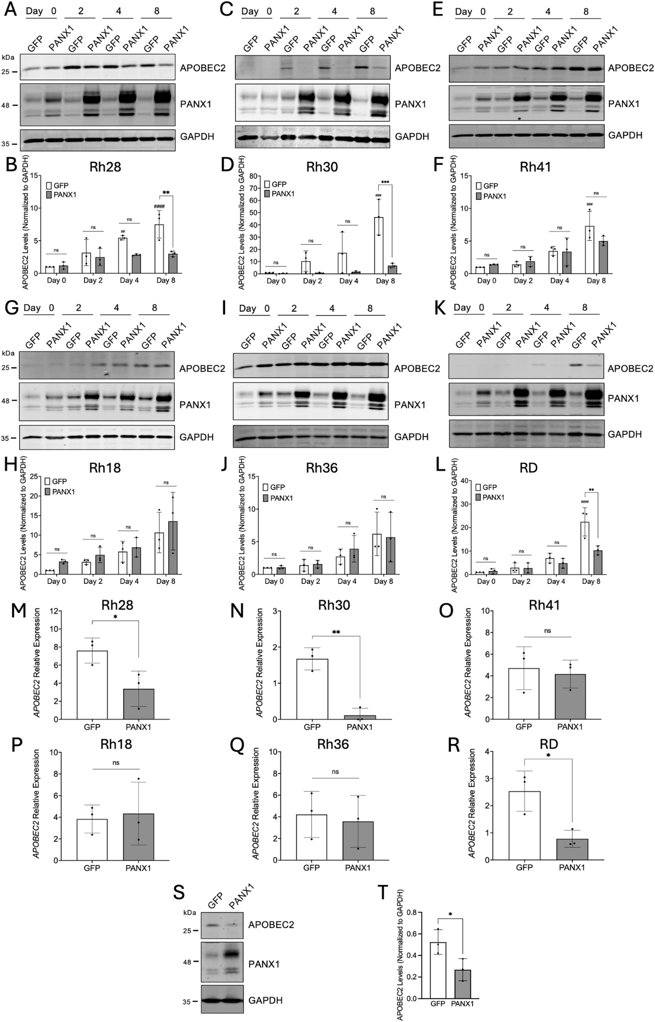
APOBEC2 levels were analyzed over eight days in our stable RMS cell lines in which PANX1 was overexpressed under the control of a cumate switch system. APOBEC2 levels increased in most control cells as they proliferate, which was significant after eight days in Rh28, Rh30, Rh41, and RD cells (Fig. 1A-L). Notably, PANX1 overexpression significantly decreased the expression of APOBEC2 in Rh28, Rh30, and RD cells compared to their respective controls but not in Rh36, Rh41 and Rh18 (Fig. 1A-L). The decrease in APOBEC2 protein levels in Rh28, Rh30 and RD cells was similarly associated with a significant decrease in its mRNA expression (Fig. 1M, N, R). Accordingly, APOBEC2 transcript expression was not affected by the upregulation of PANX1 expression in Rh41, Rh18, and Rh36 cells (Fig. 1O, P, Q). Western blot analysis showed a downregulation of APOBEC2 levels when PANX1 expression was increased in Rh30 xenografts (Fig. 1S, T), suggesting that this regulation also occurs in vivo. These results indicate that, in some RMS cell lines, increasing PANX1 levels leads to a downregulation of APOBEC2 expression at both the transcript and protein level.
Ask a Question
Write your own review
- You May Also Need
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells