
OE33
Cat.No.: CSC-C9231W
Species: Homo sapiens (Human)
Source: Esophagus
Morphology: epithelial
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
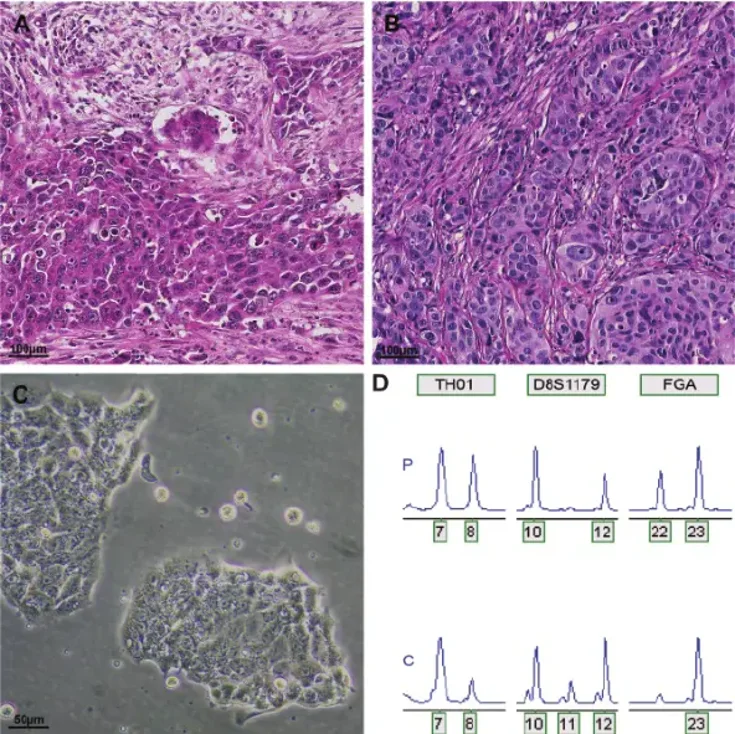
OE33 is a human esophageal adenocarcinoma cell line, originally isolated from a poorly differentiated adenocarcinoma located in the lower portion of the esophagus. The cell line has been used extensively as an in vitro tumor model system for studying the biology of esophageal adenocarcinoma (EAC), including tumor progression and Barrett's esophagus-associated carcinogenesis. OE33 cells exhibit epithelial morphology and form an adherent monolayer when maintained under typical cell culture conditions.
On a molecular level, OE33 cells share many of the genetic alterations found in esophageal adenocarcinoma, including TP53 mutations and genomic copy number alterations that regulate cell proliferation, apoptosis, and signal transduction. Therefore, OE33 can be used as a representative model cell line to study dysregulated pathways involved in EGFR- and HER2-dependent signaling and chemotherapy/radiotherapy resistance mechanisms. These cells have been used to study tumor cell proliferation, migration and invasion, epithelial-mesenchymal transition, and drug response. The cells can also be used to generate xenografts in immunodeficient mice.
Mn Frequencies and CBPI Score in TK6 and OE33 Cells Upon Plasma Treatment
In this paper, Naser et al. studied the potential genotoxic effects of human plasma from healthy volunteers, as well as patients with gastro-oesophageal reflux disease, Barrett's oesophagus (BO) and oesophageal adenocarcinoma (OAC) using the oesophageal adenocarcinoma cell line (OE33) and the lymphoblastoid cell line (TK6).
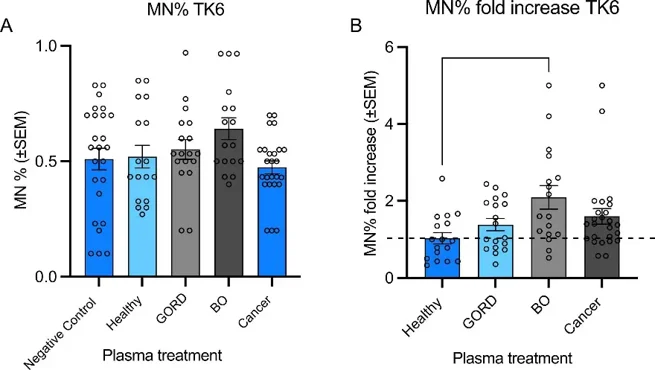
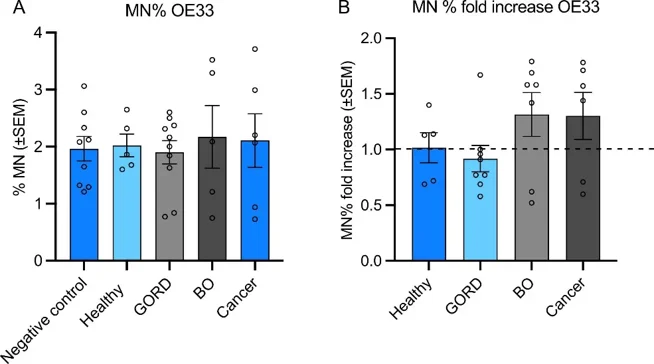
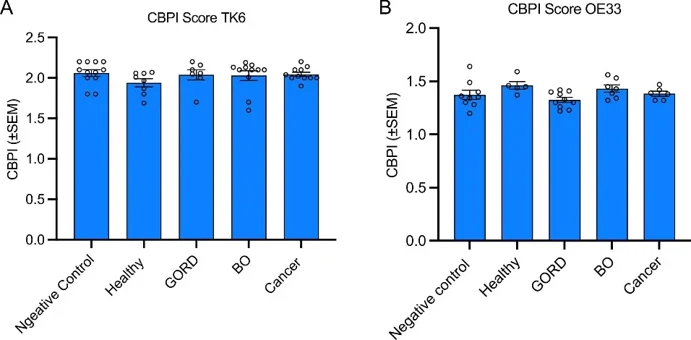
Micronucleus (Mn) frequencies were assessed in TK6 and OE33 cells after treatment with patient plasma samples. In TK6 cells (77 samples), plasma from Barrett's esophagus (BO) patients significantly increased Mn frequency versus healthy volunteers (Fig. 1B), though with considerable inter-individual variability (0.43-fold decrease to 5-fold increase). Neither donor age (p=0.4631) nor gender (p=0.3514) affected Mn production. In OE33 cells (26 samples), plasma from BO and esophageal adenocarcinoma (OAC) patients showed non-significant Mn increases (p=0.4302 and p=0.4939; Fig. 2B), with similar variability (0.62-fold decrease to 1.79-fold increase). Age (p=0.2854) and gender (p=0.3956) had no effect. Cytokinesis-block proliferation index (CBPI) was unaffected by plasma treatment in both cell lines (TK6: 37 samples; OE33: 28 samples), though individual variation existed (TK6: 1.7-2.1; OE33: 1.2-1.56; Fig. 3).
Ask a Question
Write your own review
- You May Also Need
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells





