HeLa 229
Cat.No.: CSC-C9423L
Species: Homo sapiens (Human)
Source: Uterus; Cervix
Morphology: epithelial
Culture Properties: monolayer
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Isoenzyme: G6PD, A
Production: keratin
Histopathology: adenocarcinoma
Note: This cells can be adapted to growth in suspension.; relatively resistant to polioviruses.; positive for keratin by immunoperoxidases staining
vWA: 16,18
FGA: 18,21
Amelogenin: X
TH01: 7
TPOX: 8,12
CSF1P0: 9,10
D5S818: 11,12
D13S317: 12,13.3
D7S820: 8,12
Isolated as a clonal subline from the parental HeLa strain, HeLa 229 retains the aggressive epithelial phenotype and aneuploid karyotype of its origin while exhibiting a distinctive, application‑defining functional divergence: relative insusceptibility to poliovirus coupled with superior permissiveness for obligate intracellular bacterial pathogens.
Unlike the parental line, HeLa 229 displays marked resistance to poliovirus 1, yet remains fully susceptible to human adenovirus 7 and vesicular stomatitis virus. This selective permissiveness enables controlled virological studies where poliovirus interference must be minimized.
Under standardized DEAE‑dextran/cycloheximide treatment, HeLa 229 supports high‑yield propagation of Chlamydia trachomatis and Chlamydia pneumoniae. It produces significantly greater inclusion counts and a threefold higher infectious progeny yield than the reference McCoy cell line, establishing it as the superior system for chlamydial isolation, diagnostics, and vaccine research.
HeLa 229 is documented to readily adapt to suspension culture, a trait not universally shared by adherent cervical carcinoma lines. This capability, combined with its robust doubling time (~26 h), facilitates scalable biomanufacturing and high‑throughput screening platforms.
These attributes distinguish HeLa 229 not as a generic HeLa substitute, but as a phenotypically honed tool-balancing permissiveness and resistance in ways precisely calibrated for chlamydiology, viral receptor mapping, and suspension‑adapted oncological cell engineering.
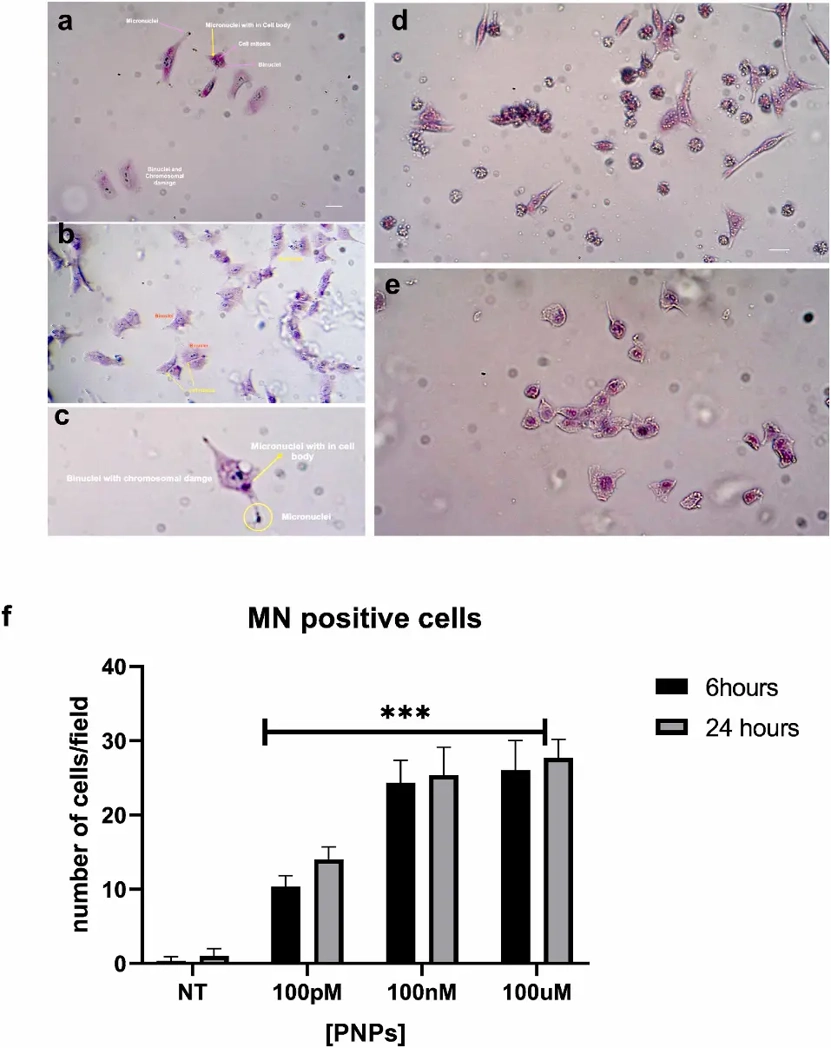
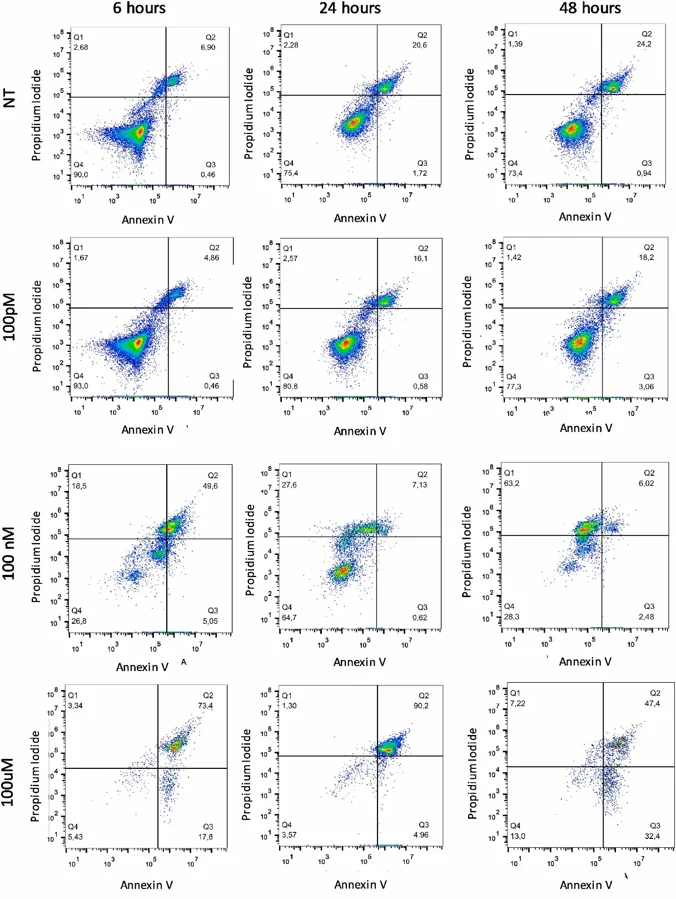
Polystyrene Nanoparticles Induce DNA Damage and Apoptosis in HeLa Cells
Polystyrene nanoplastics (PS-NPs) are the prevalent form of nanoparticles found in the environment and their cellular uptake can cause cytotoxicity and structural alteration of biomolecules. Thus, there is an urgent need for evaluation of the genotoxic effects of PS-NPs on human cell models.
Through different and complementary experimental approaches, we investigated the potential genotoxic and cytotoxic effects of PS-NPs exposure on HeLa cell lines. We highlighted the genotoxic effects of polystyrene nanoplastics by showing the formation of multinuclei and micronuclei in all the studied concentrations and time points, also at short incubation time (6 h) and low concentration. At higher concentrations, we demonstrate the presence of apoptotic and necrotic cells outlining the acute cytotoxic effects of nanoplastics. The genotoxic potential is further highlighted by the presence of low molecular weight DNA fragments in PS-NPs treated cells, and by the relationship between polystyrene nanoplastics and γ-H2AX. Our data provide important insights at a cellular level into the possible risks of these nanoparticles and recommend further investigation to address the impacts of nanoplastics on human health.
Ask a Question
Write your own review
- You May Also Need
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells