HEC-1-B
Cat.No.: CSC-C9264W
Species: Homo sapiens (Human)
Source: Uterus; Endometrium
Morphology: epithelial
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
HEC-1-B is a cell line derived from human endometrial adenocarcinoma. It was originally isolated from a patient with endometrial cancer and exhibits an endometrial carcinoma moderately differentiated phenotype. These cells have been used extensively as a model system to study endometrial cancer biology in vitro including tumor progression and response to therapeutics.
In culture, HEC-1-B cells form an adherent epithelial like monolayer with polygonal/cobblestone cell morphology. HEC-1-B cell growth has been shown to be relatively stable when compared to other endometrial cancer cell lines with fewer variations and greater reproducibility between experiments. These cells express epithelial markers and demonstrate dysregulation of pathways associated with cell cycle progression, apoptosis and hormone receptor responsiveness. Typically, HEC-1-B cells are estrogen and progesterone receptor-negative and have been used to study hormone independent growth of endometrial tumors. HEC-1-B cells have been used in the study of signal transduction, invasion and metabolism in endometrial carcinoma. HEC-1-B cells have been used as a model system for testing the efficacy of chemotherapeutics and targeted anticancer agents.
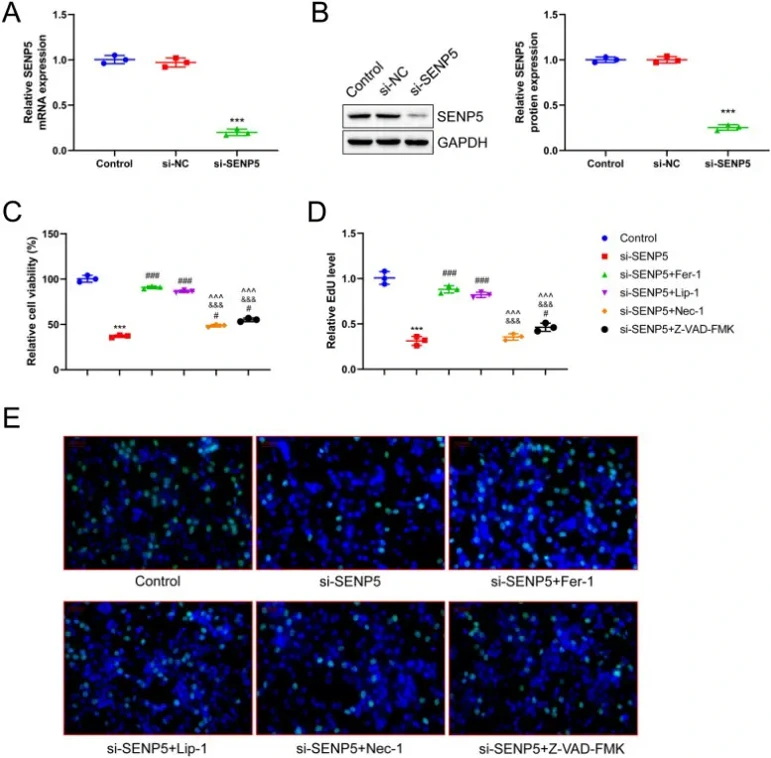
SENP5 Promotes Proliferation of EC Cells by Inhibiting Ferroptosis
Endometrial cancer (EC) poses significant risks to women's health, yet the role of SUMO specific peptidase 5 (SENP5) in EC progression remains elusive. Given ferroptosis's importance in EC development, Wang's team investigated whether SENP5 regulates EC progression through this mechanism.
SENP5 was successfully knocked down in HEC-1-B cells using siRNA, confirmed by RT-qPCR and Western blot (Fig. 1A, B). CCK-8 assays showed that SENP5 silencing significantly reduced cell viability, with ferroptosis inhibitors Fer-1 and Lip-1 substantially reversing this effect. Necrosis inhibitor Nec-1 and apoptosis inhibitor Z-VAD-FMK also partially rescued viability, but less effectively (Fig. 1C). EdU assays confirmed that ferroptosis inhibition most effectively alleviated the reduced proliferation caused by SENP5 knockdown, compared to limited effects from necrosis or apoptosis inhibition (Fig. 1D, E). These findings indicate SENP5 suppresses ferroptosis to promote EC cell proliferation.
Ask a Question
Write your own review
- You May Also Need
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells